Evaluate the integral using FTC I. L' (su + u J-1 (5u+u - u) du
Question:
Evaluate the integral using FTC I.
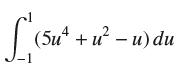
Transcribed Image Text:
L' (su² + u² J-1 (5u²+u² - u) du
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
1 1 8 5u u u ...View the full answer

Answered By

Michael Mulupi
I am honest,hardworking, and determined writer
4.70+
72+ Reviews
157+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Mathematics questions
-
Evaluate the integral using FTC I. T/6 10 sec 0 tan 0 de
-
Evaluate the integral using FTC I. WT/3 0 cost dt
-
Evaluate the integral using FTC I. -13 10 sin x dx
-
You firm needs to pay its British supplier 1,000,000. If the exchange rate is $1.61/, how many dollars will you need to pay the British supplier? OA. $1,000,000 OB. $621,118 c. $1,610,000 D. $385,787
-
What title does Tootsie Roll use for gross profit? How did it present gross profit? By how much did its total gross profit change, and in what direction, in 2009?
-
You place an order for 900 units of Good X at a unit price of $58. The supplier offers terms of 1/25, net 40. a. How long do you have to pay before the account is overdue? If you take the full...
-
Reconsider the data from Problem 56. What is the capital recovery cost of Model 127B? Data from problem 56 Octavia Bakery is planning to purchase one of two ovens. The expected cash flows for each...
-
Majestic Trucking Inc. (Majestic) is a small trucking company that carries freight between centres in central Canada and the northeastern United States. The Mozart family of Cobourg owns Majestic,...
-
One way financial managers evaluate a firm's current financial condition is by computing ratios based on current accounts listed on the firm's financial statements. Financial managers look at four...
-
Find formulas for the functions represented by the integrals. Sos sin u du
-
Write the integral in terms of u and du. Then evaluate. fox. + 1) cos(x + 4x) dx, u = x + 4x
-
Chesrown Motors is a chain of car dealerships. Sales in the fourth quarter of last year were $8,000,000. Suppose its management projects that its current year's quarterly sales will increase by 4% in...
-
Find the mean and variance of the random variable Y = X2 with X obeying the following distributions: (i) X~ N(1,0), (ii) X~ P(X), (iii) X~ exp(X).
-
It is often a challenge to evaluate purchasing because it is difficult to determine the causes of problems that might or might not be caused by the purchasing process. assume you were the manager of...
-
For the Brand "NO-Name" in Canada, If the brand is doing new campaign to boost branding and sales, How the Evaluation of the results of the communication/measurement be done? 1. Pre-testing, 2....
-
Arrays w/ User Input Objective: Create a program that allows a user to fill in values for two separate arrays, then prints the values for both arrays. Related SLOs: SLO #1: Use an appropriate...
-
Consider the following polynomial inequality. x^(2)(x+8)(x-2)>0 Step 1 of 2: Write the polynomial inequality in the form p(x) <0, p(x) 0, or p(x)>=0; then find the real zeros of p(x).
-
What are some of the control concerns in backup and recovery of data warehousing?
-
Design and describe an application-level protocol to be used between an automatic teller machine and a bank's centralized computer. Your protocol should allow a user 's card and password to be...
-
Use the Monotonicity Theorem to prove each statement if 0 < x < y. (a) x2 < y2 (b) x < y (c) 1/x > 1/y
-
What condition on a, b, and c will make f(x) = ax3 + bx2 + cx + d always increasing?
-
Determine a and b so that f(x) = ax + b / x has the point (4, 13) as an inflection point.
-
Calculate the unit cell edge length for an 79 wt% Ag- 21 wt% Pd alloy. All of the palladium is in solid solution, and the crystal structure for this alloy is FCC. Room temperature densities for Ag...
-
Calculate the energy (in eV/atom) for vacancy formation in some metal, M, given that the equilibrium number of vacancies at 418C is 6.81 x 1023 m3. The density and atomic weight (at 418C) for this...
-
Construct liquid-vapour and solid-vapour phase boundaries for iodine l2 given the following: Enthalpy of vaporization = 41.57 kJ/mol Enthalpy of fusion = 14.73 kJ/mol Normal boiling point = 386.8 K...

Study smarter with the SolutionInn App


