Let v(t) = r '(t). Show that N(t) v(t)r'' (t) - v'(t)r' (t) |v(t)r(t) - v'(t)r' (t)||
Question:
Let v(t) = ΙΙr '(t)ΙΙ. Show that
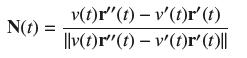
N is the unit vector in the direction T'(t). Differentiate T(t) = r'(t)/v(t) to show that v(t)r"(t) − v'(t)r (t) is a positive multiple of T '(t).
Transcribed Image Text:
N(t) v(t)r'' (t) - v'(t)r' (t) |v(t)r"(t) - v'(t)r' (t)||
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Since vt r t and Tt is a unit vector in the direction at r t we may write r t ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Mathematics questions
-
Suppose variables x,y denote people. Y(x,y) denotes "x is younger than y". F (x,y) denotes "x is a friend of y". Translate the following sentences into logic: a. Emma is the youngest person b. Ruth...
-
Lawson Consulting had the following accounts and amounts on December 31. The Retained Earnings account balance at December 1 was $0 and the owner invested $16,070 cash in the company on December 1....
-
In matlab please. Array access and modification Perform all the following operations in a script using array operations! Make sure you document it properly and minimize the # of operations. Each of...
-
1. Calculate the density of O2, in g/L, at STP. 1.62 g/L b. 0,714 g/L 1.43 g/L. d. 1.14 g/L e. 0.810 g/L a. .
-
What is meant by a dilutive security?
-
1. Ignore the test. Sheryl has proven herself via work experience and deserves the job. 2. Give the job to the candidate with the highest score. You don't need to make enemies on the Civil Service...
-
What are the major functions of marriage and the family?
-
Refer to your results from P22-22A, P22-23A, and P22-24A. Assume the following changes to the original facts: a. Collections of receivables are 60% in the month of sale, 38% in the month following...
-
With an interest of $ 2 4 . 3 1 and a principal of $ 3 , 5 0 0 for 1 0 0 days, using the ordinary interest method, the rate is ?
-
Find the normal vector to the clothoid (Exercise 35) at t = 1/3 . Data From Eexercie 35 Plot the clothoid r(t) = (x(t), y(t)), and compute its curvature (t) where = f*sin", du. x(t) = y(t) = f cos...
-
Use Eq. (12) to find N at the point indicated.
-
Below is a list of categories for sustainability costs (Exhibit 17.9). Level 1 Conventional costs Level 2 Hidden costs Level 3 Contingent costs Level 4 Image and relationship costs Level 5 ...
-
If a company behaves unethically or illegally, such as Amazons treatment of their employees, or in their marketing, such as Volkswagens falsifying their clean diesel claims, discuss how this behavior...
-
Suppose you are a huge fan of Starbucks coffee.You use the mobile app to order online and earn free rewards, you tweet about your experience with baristas and your reactions to new flavors, and you...
-
Chris White owns Soup & Salad Caf. The terms of the last order for produce were 3/15 net 60.The total amount of the order is \($1,500.\) How much will Chris pay if payment is made at the end of 60...
-
The local small appliance store has microwave ovens for sale at a price of \($149.95.\) The price tag notes that the manufacturers suggested retail price is \($160.\) This type of pricing is referred...
-
Why should a customer be concerned about transit inventory cost if they pay for the inventory only when the merchandise arrives at their premises?
-
The following are selected 2014 transactions of Palmeiro Corporation. Sept. 1 Purchased inventory from Ripken Company on account for $125,000. Palmeiro records purchases gross and uses a periodic...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
A store sells cashews for $5.00 per pound and peanuts for $1.50 per pound. The manager decides to mix 30 pounds of peanuts with some cashews and sell the mixture for $3.00 per pound. How many pounds...
-
A recently retired couple needs $12,000 per year to supplement their Social Security. They have $150,000 to invest to obtain this income. They have decided on two investment options:AA bonds yielding...
-
With a tail wind, a small Piper aircraft can fly 600 miles in 3 hours. Against this same wind, the Piper can fly the same distance in 4 hours. Find the average wind speed and the average airspeed of...
-
A $100,000 portfolio's returns are expected to be normally distributed, with an annual geometric return of 10% and an annualized standard deviation of 20%. Assuming +/-1.65describes 90% of the...
-
Question 11 (5 points) Saved (6.2) When reading a research article, what specifically should you be reviewing or questioning regarding the sample?
-
Explain key components and functions of money market in Malaysia.

Study smarter with the SolutionInn App


