Figure P 41.42 shows a few energy levels of the mercury atom. a. Make a table showing
Question:
Figure P 41.42 shows a few energy levels of the mercury atom.
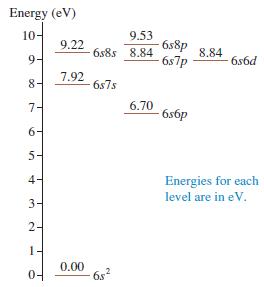
a. Make a table showing all the allowed transitions in the emission spectrum. For each transition, indicate the photon wavelength, in nm.
b. What minimum speed must an electron have to excite the 492-nm-wavelength blue emission line in the Hg spectrum?
Transcribed Image Text:
Energy (eV) 10- 9.53 6s8p 6s7p 9.22 6s8s 8.84 8.84 9- 6s6d 8- 7.92 6s7s 7- 6.70 6s6p 6- 5- 4- Energies for each level are in eV. 3- 2- 1- 04 0.00 6s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Visualize Solve a We need to use the condition l 1 to ...View the full answer

Answered By

Surbhi kapoor
Finance has always been a subject of my interest. I have been indulged in helping my classmates with this subject right from the beginning of my CA cirriculum. Helping students gives me immense amount of satisfaction. Also as I have been helping my friends from a long time with this subjects of finance, accounting and cost accounting, it has developed me to make myself more expressive about the concepts of subjects. And I also feel that I understand the problem area in a particular question. This quality makes me to better understand the student's confusion and try to answer correctly.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physics For Scientists And Engineers A Strategic Approach With Modern Physics
ISBN: 9780321740908
3rd Edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Mathematics questions
-
Figure P 41.41 shows the first few energy levels of the lithium atom. Make a table showing all the allowed transitions in the emission spectrum. For each transition, indicate a. The wavelength, in...
-
The first three energy levels of the fictitious element X were shown in FIGURE P38.56. An electron with a speed of 1.4 10 6 m/s collides with an atom of element X. Shortly afterward, the atom emits...
-
The first three energy levels of the fictitious element X are shown in FIGURE P38.56. a. What is the ionization energy of element X?b. What wavelengths are observed in the absorption spectrum of...
-
From the densities of the lines in the mass spectrum of krypton gas, the following observations were made: Somewhat more than 50% of the atoms were krypton-84. The numbers of krypton-82 and...
-
The average atmospheric pressure on earth is approximated as a function of altitude by the relation Patm = 101.325 (1 - 0.02256z)5.256, where Patm is the atmospheric pressure in kPa and z is the...
-
3. A semi-circular wire is formed with the help of a wire of length l. Find the position of the center of mass of the semicircular wire from the point "O" as shown in the figure R
-
Find the area under the standard normal curve between z = 1.5 and z = 1.25.
-
An amusement park, whose customer set is made up of two markets, adults and children, has developed demand schedules as follows: The marginal operating cost of each unit of quantity is $5. (Hint:...
-
In what ways is servant leadership a paradox? Is servant leadership more of a trait or behavior 250 words ?
-
a) ABC gross payroll for April is $65000. The company deducted $3600 for CPP, $1000 for EI and $15000 for income taxes from the employee cheques. What is ABCs contribution to CPP? b) ABC gross...
-
Suppose you put five electrons into a 0.50-nm-wide one dimensional rigid box (i.e., an infinite potential well). a. Use an energy-level diagram to show the electron configuration of the ground state....
-
The ionization energy of an atom is known to be 5.5 eV. The emission spectrum of this atom contains only the four wavelengths 310.0 nm, 354.3 nm, 826.7 nm, and 1240.0 nm. Draw an energy-level diagram...
-
Elections can be emotional events. After all, they may put into power a person or party whose policies may be different from those to which we've become accustomed. Political decisions affect...
-
What is the most uncertain variable in MMCs capitated plan pricing analysis?
-
How do provider incentives differ when the provider moves from a fee-for-service to a capitated environment?
-
a. What cost structure is best when a provider is primarily capitated? Explain. b. What cost structure is best when a provider is reimbursed primarily by fee-for-service? Explain.
-
a. What is scenario analysis as applied to pricing decisions? b. Why is it such an important part of the process?
-
What is the role of accounting information in pricing decisions?
-
A solid circular disk has a mass of 1.2 kg and a radius of 0.16 m. Each of three identical thin rods has a mass of 0.15 kg. The rods are attached perpendicularly to the plane of the disk at its outer...
-
Question 2 For an n x n matrix A = form) via (aij)
-
You are given the equation used to solve a problem. For each of these, you are to a. Write a realistic problem for which this is the correct equation. b. Draw the before-and-after pictorial...
-
You are given the equation used to solve a problem. For each of these, you are to a. Write a realistic problem for which this is the correct equation. b. Draw the before-and-after pictorial...
-
A pendulum is formed from a small ball of mass m on a string of length L. As FIGURE CP10.69 shows, a peg is height h = L/3 above the pendulums lowest point. From what minimum angle θ...
-
Suppose a male member currently aged 35, entered service at his age of 25, has a current salary of $75, 000 and total past salary of $650, 000. Assume that salaries increase at the beginning of each...
-
The Acme Rocket Car Company has a $9000 note due in 5 years. How much should be deposited at the end of each quarter in a sinking fund to pay off the note if the interest rate is 6.3%.
-
If you put $100 per month in account earning 3% annual interest, compounded monthly for 10 years, how much can you withdraw from that account each month for the next year?

Study smarter with the SolutionInn App


