Figure Q14.8 shows a velocity-versus-time graph for a particle in SHM. a. What is the phase constant
Question:
Figure Q14.8 shows a velocity-versus-time graph for a particle in SHM.
a. What is the phase constant Φ? Explain.
b. What is the phase of the particle at each of the three numbered points on the graph?
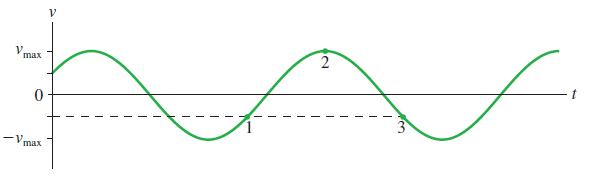
Transcribed Image Text:
max 3 -v. max 2.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
One expression for the total energ...View the full answer

Answered By

Morgan Njeri
Very Versatile especially in expressing Ideas in writings.
Passionate on my technical knowledge delivery.
Able to multitask and able to perform under pressure by handling multiple challenges that require time sensitive solution.
Writting articles and video editing.
Revise written materials to meet personal standards and satisfy clients demand.
Help Online Students with their course work.
4.90+
12+ Reviews
38+ Question Solved
Related Book For 

Physics For Scientists And Engineers A Strategic Approach With Modern Physics
ISBN: 9780321740908
3rd Edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Mathematics questions
-
FIGURE Q15.4 shows a position-versus-time graph for a particle in SHM. a. What is the phase constant Ï 0 ? Explain. b. What is the phase of the particle at each of the three numbered points on...
-
Figure a is a partial graph of the position function x(t) for a simple harmonic oscillator with an angular frequency of 1.20 rad/s; Figure b is a partial graph of the corresponding velocity function...
-
What is the phase constant for the harmonic oscillator with the velocity function v(t) given in Figure if the position function x(t) has the form x = x- cos (wt + )? The vertical axis scale is set by...
-
Which of the following options are available for creating a policy in Qualys Policy Compliance? (Choose three) A, Create from Host B, Create from Scratch C, Import from Library D, Import from CSV File
-
An ideal vapor compression refrigeration cycle with R-134a as the working fluid operates between the pressure limits of 120 kPa and 700 kPa. The mass fraction of the refrigerant that is in the liquid...
-
Consider the three-stage nested design shown in Figure 13-5 to investigate alloy hardness. Using the data that follow, analyze the design, assuming that alloy chemistry and heats are fixed factors...
-
The colour of the flame of halide torch in case of leakage of freon refrigerant, will be (a) bright green (b) yellow (c) red (d) orange
-
At the beginning of the fiscal year, Andy Company purchased for $2,060,000 a patent that applies to the manufacture of a unique tamper-proof lid for medicine bottles. Andy incurred legal costs of...
-
S Oslo Company prepared the following contribution format income statement based on a sales volume of 1,000 units (the relevant range of production is 500 units to 1,500 units): Sales Variable...
-
The New York Times cost $0.15 in 1970 and $2.00 in 2009. The average wage in manufacturing was $3.23 per hour in 1970 and $20.42 in 2009. a. By what percentage did the price of a newspaper rise? b....
-
Equation 14.25 states that 1/2kA 2 = 1/2m(v ma x ) 2 . What does this mean? Write a couple of sentences explaining how to interpret this equation.
-
Estimate the number density of gas molecules in the earths atmosphere at sea level.
-
Given her evaluation of current economic conditions, Ima Nutt believes there is a 20 percent probability of recession, a 50 percent chance of continued steady growth, and a 30 percent probability of...
-
Total cost is the sum of ____. a) marginal cost and fixed cost b) marginal cost and variable cost c) variable cost and fixed cost
-
The TV networks in the late 1940s, Microsoft in the 1990s, and Apple, most recently, have all created ecosystems of thousands of dependent fi rms, while raising a strong barrier to competition, which...
-
Greece's usage of fiscal policy to avoid the meltdown and the debt crisis As a result of the combined effects of the global financial crisis and the sovereign debt crisis, the GDP of Greece declined...
-
Give an example of a bank balance sheet with a leverage ratio of 20. If the value of the banks assets rises by 2 percent, what happens to the value of the owners equity in this bank? How large a...
-
Suppose that in a given economy, both the savings rate and the depreciation rate increase. What would the effect of this dual movement be on the economy's capital per worker and output per worker in...
-
In a nucleus, each proton experiences a repulsive electrostatic force from each of the other protons. In a nucleus of gold 197 79 Au, what is the magnitude of the least possible electrostatic force...
-
Use the graphs of f and g to graph h(x) = (f + g) (x). To print an enlarged copy of the graph, go to MathGraphs.com. 1. 2. y 24 8. 2. -2 -2 4 6
-
A 90 kg firefighter needs to climb the stairs of a 20-m-tall building while carrying a 40 kg backpack filled with gear. How much power does he need to reach the top in 55 s?
-
A 90 kg firefighter needs to climb the stairs of a 20-m-tall building while carrying a 40 kg backpack filled with gear. How much power does he need to reach the top in 55 s?
-
A hydroelectric power plant uses spinning turbines to transform the kinetic energy of moving water into electric energy with 80% efficiency. That is, 80% of the kinetic energy becomes electric...
-
A scuba diver is exploring a shipwreck at a depth of 30 meters in the ocean. The diver's tank contains 5.0 moles of compressed air at a temperature of 25C. What is the pressure of the air in the tank...
-
1. Determine the atmospheric pressure at a location where the barometric reading is 720 mm Hg. 2. A pressure gauge connected to a tank reads 52 psi at a location where the barometric reading is 29.6...
-
Assume that a bond has an annual coupon rate of 6.80 percent, but makes coupon payments on a semi-annual basis, has a par value of $1,000, a yield to maturity of 7.20 percent, and a current price of...

Study smarter with the SolutionInn App


