Given that find the matrix A 2 and, hence, show that A(A 5I) = 21, where
Question:
Given that
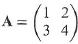
find the matrix A2 and, hence, show that A(A – 5I) = 21, where I is the identity matrix. From this equation show that the inverse matrix
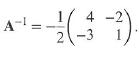
Transcribed Image Text:
(1 2) A = 3 4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)

Answered By

HARINATH G
I am a Engineering student from a prominent college of Kerala. As a tutor it's my pleasure to share my knowledge.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS & A Level Further Mathematics Coursebook
ISBN: 9781108403375
1st Edition
Authors: Lee Mckelvey, Martin Crozier
Question Posted:
Students also viewed these Sciences questions
-
Show that R -1 (a)R(a) = I, where I is the identity matrix and R(a) is the rotation matrix. This equation shows that the inverse coordinate transformation returns you to the original coordinate...
-
You are given the matrices Evaluate BICIA, where I is the identity matrix. 0 -3 0 -1 andC = 5 9 -3 -2 1 1 8 3 6 0 0 -1 0, A = 4 B = 1 7 3 4.
-
Find the inverse of the given matrix (if it exists) using Theorem 3.8. 1 1
-
The following information is available for HTM Corporation's defined benefit pension plan: On January 1, 2017, HTM Corp. amended its pension plan, resulting in past service costs with a present value...
-
Use the same televised speech/interview from the previous activity and comment on the speakers audibility, articulation, pitch, rate of speech, use of pauses, and ums and ers. Consider whether such...
-
A large art gallery has in inventory more than 100 paintings. No two are alike. The least expensive is priced at more than $1,000 and the higher-priced items carry prices of $100,000 and more. Which...
-
When a dielectric slab completely fills the space between the plates of a parallel-plate capacitor, the magnitude of the bound charge is one-fourth the magnitude of the free charge. (a) What is the...
-
Baber, Inc., manufactures custom scaffolding used in construction projects. The following data pertain to its operations for the most recent year: Raw materials beginning inventory ... $23,000 Raw...
-
22 22 The atomic number of an element T is 10. T shows similar properties to an element with atomic number 23. 24. A. 14 B. 16 C. 18 d. 20 The electron configurations of elements W, X, Y and Z are...
-
Capstone Case: Sunrise Bakery Expansion The Sunrise Bakery Corporation was originally founded in Houston, TX in 1991 by Griffin Harris, who currently serves as the company's Chief Executive Officer....
-
You are given the matrix Show that the row operation r 1 cr 1 ar 3 changes the determinant by a factor of c. ad g be h A = le f i fi)
-
Given that and calculate the matrices: a. 2A + 5B b. AB 2 c. BA 2 d. 2A + 3A 2 1 2 -5 9 -1 -5 10, A = 3
-
Explain how accounting practice defines a product financing arrangement.
-
1. Given the IP address of 172.16.1.1 with a mask of 255.255.255.0-- How many total subnets could be created? (assume all subnets use the same subnet mask) a. 65536 b. 254 c. 256 d. 64 2. Represent...
-
What would you/your group consider you to be an extrinsic source on? What would you be considered an intrinsic source on? My group would come to me for all sorts of parenting ideas and thoughts,...
-
How to Calculate taxable income by reducing gross income by adjustments, exemptions, and standard or deductions?
-
Gavin is the disaster recovery team leader for his organization, which is currently in the response phase of an incident that has severe customer impact. Gavin just received a phone call from a...
-
Transition Planning Team: Who are the stakeholders and what are their roles and responsibilities? Families are an essential part of the transition team. How will the transition team ensure that the...
-
A ray of light passes from air through dense flint glass and then back into air. The angle of incidence on the first glass surface is 60.0. The thickness of the glass is 5.00 mm; its front and back...
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Identify the major and minor products for each of the following E1 reactions: a. b. c. d. H,SO. eat ETOH Heat Br
-
Identify two different starting alcohols that could be used to make 1-methylcyclohexene. Then determine which alcohol would be expected to react more rapidly under acidic conditions. Explain your...
-
Draw only the major product for each of the following E1 reactions: a. b. H,SO, Heat H2SO. eat
-
Cash Balance July 1, 2024 Cash Receipts $6,990 + $8,330 RAVINE INCORPORATED Cash Account Records July 1, 2024, to July 31, 2024 Cash Disbursements Cash Disbursements Date 7/9 7/21 7/31 Cash Balance...
-
College Pizza delivers pizzas to the dormitories and apartments near a major state university. The company's annual fixed expenses are $57,000. The sales price of a pizza is $10, and it costs the...
-
Sweeten Company had no jobs in progress at the beginning of the year and no beginning inventories. It started, completed, and sold only two jobs during the year-Job P and Job Q. The company uses a...

Study smarter with the SolutionInn App


