A cell with a water potential of 300 kPa was placed in pure water at time zero.
Question:
A cell with a water potential of –300 kPa was placed in pure water at time zero. The rate of entry of water into the cell was measured as the change in water potential with time. The graph shows the results of this investigation.
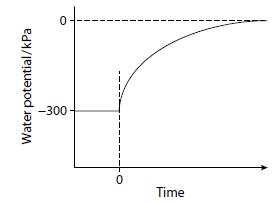
Describe and explain the results obtained.
Transcribed Image Text:
-300 Time Water potential/kPa
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
Water potential of pure water is zero Water potential of water decreases as solutes are added As mor...View the full answer

Answered By

Keerthika K
I have been the topper of my class in undergraduate and postgraduate studies. I also have immense online tutoring experience where I help students in understanding their concepts in a crystal clear manner. I am also strongly analytical which helps me answer every question that requires critical thinking.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Biology
ISBN: 9781107636828
4th Edition
Authors: Mary Jones, Richard Fosbery, Jennifer Gregory, Dennis Taylor
Question Posted:
Students also viewed these Sciences questions
-
At time zero the price of a non-dividend-paying stock is S 0 . Suppose that the time interval between 0 and T is divided into two subintervals of length t 1 and t 2 . During the first subinterval,...
-
Water initially at 300 kPa and 0.5 m3/kg is contained in a piston-cylinder device fitted with stops so that the water supports the weight of the piston and the force of the atmosphere. The water is...
-
A mass of 5 kg of saturated water vapor at 300 kPa is heated at constant pressure until the temperature reaches 200C. Calculate the work done by the steam during this process.
-
How many objects are eligible for garbage collection at the end of the main() method? A. None. B. One. C. Two. D. Three. E. The code does not compile. F. None of the above. package store; public...
-
The blue line of the strontium atom emission has a wavelength of 461 nm. What is the frequency of this light? What is the energy of a photon of this light?
-
Repeat Prob. 380 with Fx = 75 lbf, Fy = 200 lbf, and Fz = 100 lbf. Repeat Prob. 380, The cantilevered bar in the figure is made from a ductile material and is statically loaded with Fy = 200 lbf and...
-
Assume the expected inflation rate in China to be 4.5 percent. If the current real rate of interest is 6.5 percent, what is the nominal rate of interest?
-
SHL Americas provides a unique, global perspective of how talent is measured in its Global Assessment Trends Report. The re-port presents the results of an online survey conducted in late 2012 with...
-
6. To a ground observer the block C is moving with v, and the block A with v. B is moving with v relative to C as shown in the figure. Identify the correct statement. (a) V-V =Vo (b)= A C B (c) V+V =...
-
Figure 4-36 includes an EER diagram describing a car racing league. Transform the diagram into a relational schema that shows referential integrity constraints (see Figure 4-5 for an example of such...
-
Copy and complete the table below to compare cell walls with cell membranes. Feature Cell wall Cell membrane is the thickness normally measured in nm or um? location chemical composition permeability...
-
The rate of movement of molecules or ions across a cell surface membrane is affected by the relative concentrations of the molecules or ions on either side of the membrane. The graphs below show the...
-
Determine whether f is continuous at c. 3 cos x if x < 0 if x = 0 c = 0 3 f(x) = x3 + 3x? if x > 0
-
5. Find the volume of revolution formed by rotating the curve y = x + x, the x-axis and the ordinates x =2 and x = = 3. = 2
-
There are eight characteristics are listed for Traditional Project Management, and eight characteristics are listed for Agile Project Management. Please answer the following: Given our perspective of...
-
Part One Prepare your own lessons learned report (1-2 pages, single-spaced) based on personal reflections of what you learned from this class, including the team projects and presentations. Do not...
-
In order to keep your core values aligned with your organization's mission and goals, it's essential to reevaluate them frequently. The easiest way to do is by asking your team for feedback. From...
-
You have just completed a full report to the Board of Directors on the status and progress of your early childhood center. The report reveals that there is a high rate of turnover among the teaching...
-
The following transactions were completed by Winklevoss Inc., whose fiscal year is the calendar year: 2016 July 1. Issued $74,000,000 of 20-year, 11% callable bonds dated July 1, 2016, at a market...
-
Les has collected stamps in his spare time for years. He purchased many of his stamps at a price much lower than the current market value. Les recently lost his job as a carpenter. Since his wife...
-
Identify the reagents you would use to convert 2-bromo-2-methylbutane into 3-methyl-1-butyne.
-
Would ethanol (CH 3 CH 2 OH) be a suitable solvent in which to perform the following proton transfer? Explain your answer: ONH2 + NH3 NH2 H.
-
Identify the reagents you would use to convert 1-pentene into a geminal dibromide (geminal indicates that both bromine atoms are connected to the same carbon atom).
-
Simplify the expression with the rational exponent. 3 16. =-35/2 -162 17. (-16) 18. 32 -64 -3 = 46/3 162 Simplify. +8 19. =X10 X-2
-
This is my work please can you help disabled student word count is not Important. please help Part 2 For this part of the assignment, you should write an answer to the following question: What can...
-
Suppose that x is the fraction of UCSC students who prefer calculus to linear algebra at year k. The remaining fraction y = 1-x prefers linear algebra. 1 '5' of those who prefer calculus change their...

Study smarter with the SolutionInn App


