The graph shows the eff ect of changes in pH on the activity of the enzyme lysozyme.
Question:
The graph shows the eff ect of changes in pH on the activity of the enzyme lysozyme.
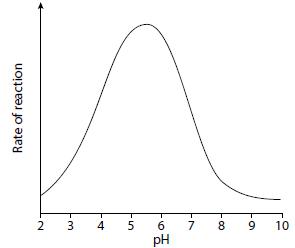
a. Describe the effect of pH on this enzyme.
b. Explain why pH affects the activity of the enzyme.
Transcribed Image Text:
4 6. 7 8. 10 pH 9, 2. Rate of reaction
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
A Effect of ph on enzyme activity as it is visible in the graph that as the ...View the full answer

Answered By

SHOAIB AHMED
I am a doctor by profession and a part time tutor having experience with some famous sites like Chegg , Bartleby, and course hero , And still working with them apart from that I have given tution classes for NEET , MDCAT students
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Biology
ISBN: 9781107636828
4th Edition
Authors: Mary Jones, Richard Fosbery, Jennifer Gregory, Dennis Taylor
Question Posted:
Students also viewed these Sciences questions
-
The graph below shows the eff ect of temperature on the rate of reaction of an enzyme. a. What is indicated by X? b. What temperature would X be for a mammalian enzyme? c. Explain what is happening...
-
Explain why enzyme activity varies with temperature, as shown here. 40 20 Temperature ("C)
-
Explain why lysozyme cleaves the artificial substrate (NAG)4 ~4000 times more slowly than it cleaves (NAG)6.
-
In Rev. Rul. 81-301, which of the following is not a factor considered by the IRS in determining whether a partner receives payments in her capacity as a partner, or is instead acting as a third...
-
What is the wavelength of the transition from n = 5 to n = 3 for Li2+? In what region of the spectrum does this emission occur? Li2+ is a hydrogen-like ion. Such an ion has a nucleus of charge +Ze...
-
For the knuckle joint described in Prob. 340, assume the maximum allowable tensile stress in the pin is 30 kpsi and the maximum allowable shearing stress in the pin is 15 kpsi. Use the model shown in...
-
Prepare an income statement and a common-sized income statement from the following information about the accounts of the Uzbekistan Timber Corporation (in Uzbekistani som). Sales Cost of goods sold...
-
Natalie Koebel spent much of her childhood learning the art of cookie-making from her grandmother. They passed many happy hours mastering every type of cookie imaginable and later creating new...
-
6. For which of following reversible reaction the degree of dissociation or association depends upon the value of temperature but NOT on value of pressure? (a) N2(g)+3H2(g)2NH3(g) (b) N2O4(g) 2NO2(g)...
-
SkiCo, Inc., manufactures ski boots. The companys projected income for the coming year, based on sales of 160,000 units, is as follows: Required: In completing the following requirements, ignore...
-
Copy the graph in question 3 and draw a line from which the initial rate of reaction could be calculated. Time Amount of product formed
-
The reaction below occurs during aerobic respiration. The reaction is catalysed by the enzyme succinate dehydrogenase. a. Name the substrate in this reaction. b. The molecule malonic acid, which is...
-
Identify the two major types of clostridial food poisoning. Which is most prevalent? Which is most dangerous and why?
-
If you pay $ 2 0 0 0 at the beginning of each year for 1 0 years for your insurance scheme which earns 1 2 % compounded annually, what is the present value of your insurance scheme?
-
In February 1966, your father purchased a Pete Rose rookie edition baseball card for $0.60. As is the tradition with baseball cards, your father also received a small, thin piece of gum that quickly...
-
21D. A wave equation (in SI) is y = 0.12 sin(1.5t - 0.67x). (a) What is the amplitude? (b) What is the frequency? (c) What is the wavelength? (d) What is the displacement at x = 2.5 m when t = 6.5 s?
-
2. In 123.456, the hundreds place is the third place to the left of the decimal point; is the hundredths place the third place to the right of the decimal point? In a long numeral like 333331.333333,...
-
You deposit $ 1 , 2 0 0 in an account each year for 5 years, starting at the end of Year 1 . At the end of Year 5 , you withdraw half of the account balance. You then deposit $ 3 , 3 0 0 each year...
-
On February 10, 15,000 shares of Sting Company are acquired at a price of $25 per share plus a $150 brokerage commission. On April 12, a $0.40-per-share dividend was received on the Sting Company...
-
Which of the following statements is false? a. Capital leases are not commonly reported in a Capital Projects Fund. b. A governmental entity may report a Capital Project Fund in one year but not the...
-
The rate at which two methyl radicals couple to form ethane is significantly faster than the rate at which two tert-butyl radicals couple. Offer two explanations for this observation.
-
There are only two stereo-isomers of 1, 4-dimethylcyclohexane. Draw them, and explain why only two stereo-isomers are observed.
-
Syn dihydroxylation of the compound below yields two products. Draw both products and describe their stereo-isomeric relationship (i.e., are they enantiomers or diastereomers?): MO,, NaOH Cold
-
e The following table exhibits the average weights (in grams) of 4 randomly selected soap bars for 6 consecutive days. Assuming that the production process has a standard deviation of 2.5, answer the...
-
Please identify 2 pros (advantages) and 2 cons (disadvantages) of investing in real estate as a Private Equity Investor within a syndicate or consortium of equity investors (that is, you are an...
-
A 100-Year Bond does not have appreciably more price risk than a 30-Year Bond. Why is this?

Study smarter with the SolutionInn App


