a. Draw a diagram of an electrochemical cell consisting of a Mn 2+ /Mn half-cell and a
Question:
a. Draw a diagram of an electrochemical cell consisting of a Mn2+/Mn half-cell and a Pb2+/Pb half-cell.
b. Use the data in Appendix 2 to calculate the cell voltage.
c. Which half-cell is the positive pole?
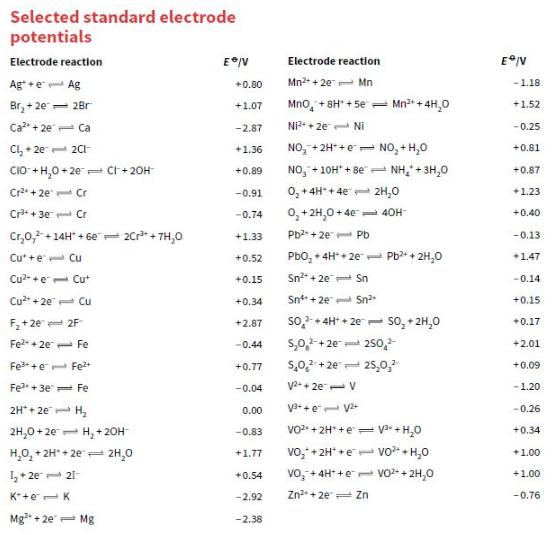
Transcribed Image Text:
Selected standard electrode potentials Electrode reaction Electrode reaction Ag+ +e- Ag Mn2 + 2e- Mn +0.80 -1.18 Br, + 2e= 28r Mno, + 8H* + 5e = Mn2* + 4H,0 +1.07 +1.52 Ca + 2e Ca -2.87 Ni2+ + 2e- Ni - 0.25 NO, +2H* +e= NO, + H,0 +0.81 Cl, + 2e 2CH +1.36 CIO +H,0+ 2e- Cr + 20H NO, +10H" + 8e"- NH,+ 3H,0 +0.89 +0.87 Cr* + 2e Cr -0.91 0, +4H* + 4e 2H,0 +1.23 Cr+ 30 Cr 0,+ 2H,0+ 4e- 4OH -0.74 +0.40 Cr,0,+14H" + 6e= 20r* + 7H,0 +1.33 Pb2 + 2e Pb -0.13 Cu* +e Cu +0.52 Pbo, + 4H* + 2e 1 Pb* + 2H,0 +1.47 Cu2- +e Cut +0.15 Sn2* + 20 Sn -0.14 Cu + 20 Cu +0.34 Snt + 2e Sn +0.15 so + 4H* + 2e so, + 2H,0 F,+2e= 2F +2.87 +0.17 s,0,-+2e- 250, 5,0,2 + 2e= 25,0, Fe2 + 20 Fe -0.44 +2.01 Fe++e- Fe2+ +0.77 +0.09 Fe*+3e Fe -0.04 V+ 2e- V -1.20 2H* + 2e H, V3-+e V -0.26 0.00 2H,0 + 2e- H, + 20H VO2 + 2H* +e = + H,0 -0,83 +0.34 H,0, + 2H* + 2e 2H,0 vo,+ 2H* +e vo+H,0 +1.77 +1.00 1+ 2e 21- vo, + 4H* +e vo+ + 2H,0 +1.00 +0.54 K*+e-K -2.92 Zn + 2e Zn -0.76 Mg + 2e- Mg -2.38
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
a Im sorry I am a textbased model and am unable to draw diagrams But I can describe the basic compon...View the full answer

Answered By

FELIX NYAMBWOGI
I have been tutoring for over 5 years, both in person and online. I have experience tutoring a wide range of subjects, including math, science, English, and history. I have also worked with students of all ages, from elementary school to high school.
In addition, I have received training in effective tutoring strategies and techniques, such as active listening, questioning, and feedback. I am also proficient in using online tutoring platforms, such as Zoom and Google Classroom, to effectively deliver virtual lessons.
Overall, my hands-on experience and proficiency as a tutor has allowed me to effectively support and guide students in achieving their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
a. Draw a diagram of an electrochemical cell consisting of a Cr 3+ /Cr half-cell and a Cl 2 /Cl half-cell. b. Use the data in Appendix 2 to calculate the cell voltage. c. Which half-cell is the...
-
Use the data in Appendix 3 to calculate the equilibrium constant for the reaction Agl(s) Ag+(aq) + I2(aq) at 25C. Compare your result with the Ksp value in Table 16.2.
-
The data in Appendix D (available at www.uvm.edu/~dhowell/fundamentals8/DataFiles/Add.dat) are actual data on high school students. What is the 75th percentile for GPA in these data? (This is the...
-
You have recently been hired as a consultant for a personal financial planning firm. One of your first projects is creating a retirement plan for a couple, Tom and Helena Keeley. They have just...
-
Let z be a random variable with a standard normal distribution. Find the indicated probability and shade the corresponding area under the standard normal curve. P( 2.37 z 0)
-
A construction project is broken down into the following 10 activities: a. Draw the network diagram. b. Find the critical path. c. If activities 1 and 10 cannot be shortened, but activities 2 and 9...
-
You throw a ball straight up. Draw a free-body diagram for the ball (a) while it is still touching your hand and is accelerating upward, (b) at its highest point, (c) on the way back down.
-
An injection molding machine has a first cost of $1,050,000 and a salvage value of $225,000 in any year the machine is sold. The maintenance and operating cost is $235,000 with an annual gradient of...
-
1. What is Stock Market Analysis. Explain in Detail? 2. Discuss about Nature and Scope of Stock Market Analysis. 3. Discuss about Basics of Stock Market Analysis. 4. What is Investment? Explain in...
-
The adjusted trial balance of Boston Irrigation System at December 31, 2018, follows: Requirements 1. Prepare the company's income statement for the year ended December 31, 2018. 2. Prepare the...
-
Show, with the aid of a diagram, how you would measure the E Q value for the half-cell shown by the equation: VO 2+ + 2H + + e V 3+ + H 2 O
-
State the direction of the electron flow in the electrochemical cells represented by the following pairs of half-equations. Use the data in Appendix 2 to help you. a. F 2 + 2e 2F and Mn 2+ + 2e ...
-
A 75 kW radio transmitter emits 550 kHz radio waves uniformly in all directions. At what rate do photons strike a 1.5-m-tall, 3.0-mm-diameter antenna that is 15 km away?
-
Suppose that there is a sales job opportunity that is entirely based on commission- Income earned depend on how much you sell. There are two outcomes: the first possible outcome is "successful"...
-
A potential investment costs $2000. You estimate that the investment has three possible outcomes. 1) it is worth $1,943 (p=.25) 2) it is worth $2,216 (p=.25) 3) it is worth $3,172 (p=.5) What is the...
-
Jan wants to plan for her daughter's education. Her daughter, Rachel was born today and will go to college at age 18 for five years. Tuition is currently $15,500 per year, in today's dollars. Jan...
-
Troy Engines, Ltd., manufactures a variety of engines for use in heavy equipment. The company usually produces all of the necessary parts for its engines, including all of the carburetors. An outside...
-
Pete's General Store is a retail business. The company's year-end gross profit is $81,600, and sales are $781,500. What is its cost of goods sold?
-
A hydrogen atom in its ground state absorbs a 97-nm ultraviolet photon. It then emits one or more photons to return to the ground state. (a) If the atom is at rest before absorbing the UV photon,...
-
What are three disadvantages of using the direct write-off method?
-
Assume that air has a mean molar mass of 28.9 g mol 1 and that the atmosphere has a uniform temperature of 25.0C. Calculate the barometric pressure in Pa in Santa Fe, for which z = 7000. ft. Use the...
-
The heat capacity of solid lead oxide is given by Calculate the change in enthalpy of 1.75 mol of PbO(s) if it is cooled from 825 to 375 K at constant pressure. 44.35 + 1.47 10 in units of J K-1 mol1
-
a. Calculate S if 1.00 mol of liquid water is heated from 0.00C to 10.0C under constant pressure if C P,m = 75.3 J K -1 mol -1 . b. The melting point of water at the pressure of interest is 0.00C and...
-
Simplify 20 to the form ab. a = b = C
-
Find the x-intercepts (if any) for the graph of the quadratic functio f(x) = x = x + 16x + 51 Give your answers in exact form.
-
1. (points) Prove total correctness of the following Loop: {n > 0} i = 1; while( i

Study smarter with the SolutionInn App


