Copy and complete the table below to compare the properties of giant ionic, giant molecular, giant metallic
Question:
Copy and complete the table below to compare the properties of giant ionic, giant molecular, giant metallic and simple molecular structures.
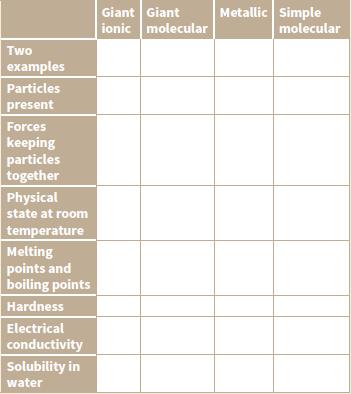
Transcribed Image Text:
Giant Giant ionic molecular Metallic Simple molecular Two examples Particles present Forces keeping particles together Physical state at room temperature Melting points and boiling points Hardness Electrical conductivity Solubility in water
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)

Answered By

Nandana Wijayarathna
I am a highly experienced writer in several areas,
Business management
Information technology
Business administration
Literature
Biology
Environmental science
History
4.50+
161+ Reviews
399+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
Copy and complete the table below to compare cell walls with cell membranes. Feature Cell wall Cell membrane is the thickness normally measured in nm or um? location chemical composition permeability...
-
Copy and complete the table at right. Then answer 3a-d to calculate the correlation coefficient. a. What is the sum of the values for (x - )(y - )? b. What are and sx? c. What are and sy? d....
-
Complete the table below for contribution margin per unit, total contribution margin , and contribution margin ratio: A Number of units 1,720 units 14,920 units 4,620 units Sales price per unit $...
-
Consider a lottery L1 = [0.5, A; 0.5, L2], where U(A) = 4, and L2 = [0.5, X; 0.5, Y] is a lottery, and U(X) = 4, U(Y) = 8. What is the utility of the the first lottery, U(L1)?
-
Express these numbers in standard notation. a. 2.87 10-8 b. 1.78 1011 c. 1.381 10-23
-
Randy Coburn opened a law office, Randy Coburn, Attorney at Law, on July 1, 2015. On July 31, the balance sheet showed Cash $4,000, Accounts Receivable $1,500, Supplies $400, Equipment $5,000,...
-
Two parallel-plate capacitors are identical except that capacitor 1 has vacuum between the plates and capacitor 2 has a dielectric slab of dielectric constant \(\kappa\) filling the space between the...
-
On April 1, 2014, Angel Corporation issued $8,000,000 in 8.5 percent, five-year bonds at 98. The semiannual interest payment dates are April 1 and October 1. Prepare journal entries to record the...
-
The price of a car you want is $39,000 today. Its price is expected to increase by $1000 each year. You now have $23,500 in an investment account, which is earning 11% per year. How many years will...
-
Below are the transactions for October 2022, for The Brown Company. Owner, Sam Smith has asked you the office manager to record the transactions into the correct journal. He has supplied you with the...
-
Suggest, using ideas of structure and bonding, why: a. Buckminsterfullerene, C 60 , is converted from a solid to a gas at a relatively low temperature b. Graphene is a good conductor of electricity...
-
Explain the following properties of silicon(IV) oxide by referring to its structure and bonding. a. It has a high melting point. b. It does not conduct electricity. c. It is a crystalline solid. d....
-
Is there any instance in which a change in tax years is required? Explain.
-
National income taxes in Germany $1,500,000 City of Munich income taxes $ 200,000 Value added tax to German government $ 400,000 Payroll tax to the German government (employer's share of social...
-
Athlete Kalen wishes to retire at age forty-five and receive annual birthday payments of $40,000 beginning on his forty-fifth birthday. After his death, the payments on the anniversary of his birth...
-
Spin Master: Canadian Toy Maker Becomes .a Global Children's Entertainment Company . Here's a business success story that will inspire you. After graduating from univensity, two young Canadian...
-
Conduct a PESTLE analysis of McDonalds. You are required to discuss the key factors that may affect its business with the use of appropriate examples.
-
Tax reliefs and deductions help to lower your chargeable income and, consequently, your income tax payable in Malaysia. Discuss with relevant examples. 1. Give 5 point and explaination above 2. Each...
-
Give the systematic name for each of the following compounds: a. b. c. d. e. f. g. h. CH2CH3 CH3 CH2CH3 H3C CH3CHCH2CH2CH3 CH3CH2CHCH3 CH3CHCH3
-
Element compound homogeneous mixture (heterogeneous mixture) 4) A piece of gold has a mass of 49.75 g. What should the volume be if it is pure gold? Gold has a density of 19.3 g/cm (3 points) D=m/v...
-
Propose a plausible synthesis for each of the following transformations: a. b.
-
In each group of compounds below, select the most acidic compound: (a) (b) Z Z CI CI CI
-
Consider the following reaction: This reaction has been determined to be second order. (a) What is the rate equation for this reaction? (b) How will the rate be affected if the concentration of...
-
The height of a nail in a bicycle tire as it rotates around is periodic in nature. The radius of the tire is 20cm and at the current pedalling speed the tire rotates once every second. a. Determine...
-
Management desires to earn approximately the same rate of profit on sales that is being earned with the current design. a. If DF uses target costing and desires to meet the current competitive...
-
How does an auditor make the following representations when issuing the standard auditors report on comparative financial statements? Examination of evidence on a test basis Consistent application of...

Study smarter with the SolutionInn App


