Use data from the table below of characteristic infra-red absorptions in organic molecules to answer the following
Question:
Use data from the table below of characteristic infra-red absorptions in organic molecules to answer the following question.
One of the three spectra labelled A to c below is produced when ethanal is analysed in an infra-red spectrophotometer:
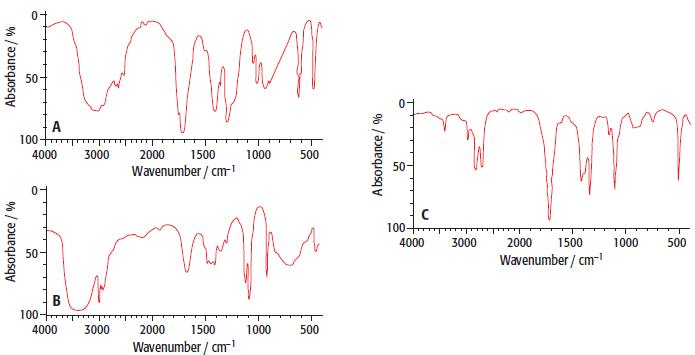
Which infra-red spectrum is most likely to be produced by ethanal? Give three reasons for your choice.
Transcribed Image Text:
Location alcohols, esters Bond Wavenumber / cm-1 C-0 1000-1300 C=0 aldehydes, ketones, carboxylic acids, esters 1680-1750 0-H hydrogen bonded in carboxylic acids 2500-3300 (broad) primary amines hydrogen bonded in alcohols, phenols N-H 3100-3500 0-H 3230-3550 0-H free 3580-3670
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Acetaldehyde another name for ethanol is a basic organic molecule having the chemical formula CH3CHO ...View the full answer

Answered By

Agrima Nigsm
In my capacity as an lecturer in biology at a school, I play a crucial role in educating students and shaping their brains to become the future generation of biologists. I am in charge of conducting research, teaching, and providing services to the school community. I have to teach as part of my job. I create and instruct classes on a variety of subjects, such as physiology, genetics, ecology, and animal behavior. I make an effort to make sure that your pupils have a solid grasp of the material and the abilities needed to utilize this knowledge in their future employment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
Use information from Table 16.2 to answer the following questions about sound in air. At 20oC the bulk modulus for air is 1.42 X 105 Pa and its density is 1.20 kg/m3. At this temperature, what are...
-
Use the following payoff matrix to answer the following questions. Suppose this is a one-shot game:a. Determine the dominant strategy for each player. If such strategies do not exist, explain why...
-
Which information below is most likely to be incorporated by reference in Form 10-K Select one: A. Controls and procedures. B. Audited financial statements and supplementary financial information. C....
-
On 1 January 2022, ABC Company issued 10,000 shares of common stock for $100,000. On 31 December 2022, the company declared and paid dividends of $10,000. Calculate the earnings per share and the...
-
What is your favorite color? A large survey of countries, including the United States, China, Russia, France, Turkey, Kenya, and others, indicated that most people prefer the color blue. In fact,...
-
Kinokawa Consultants Companys trial balance on December 31, 2014, follows. The following information is also available: a. Ending inventory of office supplies, $86 b. Prepaid rent expired, $700 c....
-
Refer to the Arctic Springs Filtration Department Data Set. 1. Compute the December conversion costs in the Filtration Department. 2. If the Filtration Department completely processed 200,000 liters,...
-
While auditing a client, the CEO asks you to look carefully at the cash flow. You notice that cash flows have decreased every year. Upon learning of your findings, the CEO remarks, I seem to bring in...
-
You are given an undirected graph consisting of n vertices and m edges. It is guaranteed that the given graph is connected (i. e. it is possible to reach any vertex from any other vertex) and there...
-
Due to a product upgrade, two new operations are need for the new version of the part mentioned in Practice Problem 1. Operation 3 is a high-precision drilling operation. Machine 3 has a mean time to...
-
Look at the two infra-red spectra below: a. Which one of the infra-red spectra is that of butanone and which one is of butan-2-ol? b. Explain your reasoning in part a. 20 20- 40 40- 60 60 80 08 100...
-
Copper(II) nitrate decomposes on heating. The reaction is endothermic. 2Cu(NO 3 ) 2 (s) 2CuO(s) + 4NO 2 (g) + O 2 (g) a. Draw an enthalpy level diagram (reaction profile diagram) for this reaction....
-
Define the concept of shopper marketing and explain why it has grown in prominence.
-
Image transcription text Role Play- SKILLS ASSESSMENT Please complete this by videoing yourself and sending the video to dised @fit edulau When undertaking your role play please be organised and...
-
Question 19 of 26 With respect to the maximum amount an individual can contribute to a TFSA, This quiz: 26 point(s) pos This question: 1 point(s) Choose the correct answer OA. any amounts that are...
-
On 1 January 20X1, YX acquired 55% of RT's equity shares. YX elected to measure non-controlling interests in RT at fair value at acquisition. In the year ended 31 December 20X3, profit for the year...
-
Shirley and Timmy form ST, an equal partnership, with Shirley contributing land held for investment with a fair market value of $93800, and an adjusted basis to her of $145200. Timmy contributes...
-
Chloe's Cafe bakes croissants that it sells to local restaurants and grocery stores. The average costs to bake the croissants are $0.45 for 2,100 and $0.40 for 4,200. Required: If the total cost...
-
In a fixed-target experiment, high-energy charged particles from an accelerator are smashed into a stationary target. By contrast, in a colliding beam experiment, two beams of particles are...
-
Evaluate the function at the given value(s) of the independent variable. Simplify the results. (x) = cos 2x (a) (0) (b) (- /4) (c) (/3) (d) ()
-
Which of the following systems are open? a) A dog, b) An incandescent light bulb c) A tomato plant d) a can of tomatoes. Explain your answers.
-
Calculate S, S surroundings , and S universe per day for the air conditioned house described in Problem 5.4. Assume that the interior temperature is 65F and the exterior temperature is 99F.
-
The following heat capacity data have been reported for L-alanine: By a graphical treatment, obtain the molar entropy of L-alanine at T = 300.K.You can perform the integration numerically using...
-
How do I post this to the ledger the town folk purchased 150 pencils by paying cash totaling $75,000?
-
Texas Manufacturing has decided to triple the number of distribution centers it operates around North America in order to locate inventories within 500 miles of each major city. What effects should...
-
Solve the problem using 6.2%, up to $128,400 for Social Security tax and using 1.45%, no wage limit, for Medicare tax.Calculate the monthly Social Security and Medicare withholdings (in $) for the...

Study smarter with the SolutionInn App


