Redo Problem 10.3-7 using Aspen Plus. Problem 10.3-7 a. Make the best estimate you can of the
Question:
Redo Problem 10.3-7 using Aspen Plus.
Problem 10.3-7
a. Make the best estimate you can of the composition of the vapor in equilibrium with a liquid containing 30.3 mol % ethane and 69.7 mol % ethylene at −0.01°C. Compare your results with the experimental data in the table.
b. Repeat the calculation in part (a) at other compositions for which the experimental data below are available.
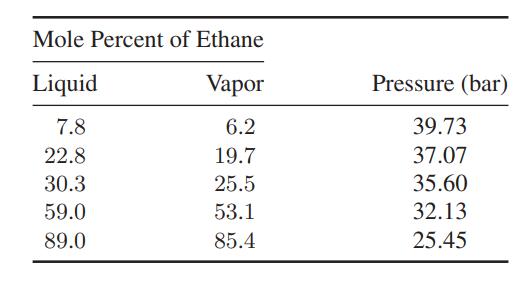
Transcribed Image Text:
Mole Percent of Ethane Liquid Vapor 7.8 6.2 22.8 19.7 30.3 25.5 59.0 53.1 89.0 85.4 Pressure (bar) 39.73 37.07 35.60 32.13 25.45
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
a. Make the best estimate you can of the composition of the vapor in equilibrium with a liquid containing 30.3 mol % ethane and 69.7 mol % ethylene at 0.01C. Compare your results with the...
-
For a separations process it is necessary to determine the VLE compositions of a mixture of ethyl bromide and n-heptane at 30C. At this temperature the vapor pressure of pure ethyl bromide is 0.7569...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
A simple beam AB of span length L = 24ft is subjected to two wheel loads acting at distance = 5 ft apart (see figure). Each wheel transmits a load P = 3.0 k, and the carriage may occupy any position...
-
A lease involves payments of $12,000 per year for five years. The payments are made at the end of each year. The lease involves a bargain purchase option of $5,000 to be exercised at the end of the...
-
The magnetic component of a polarized wave of light is given by Bx = (4.00T) sin [k y + (2.00 x 1015 s-1t]. (a) In which direction does the wave travel? (b) Parallel to which axis is it polarized,...
-
In a 1975 contract, Eureka was given the exclusive right to sell spring water and other products under the Ozarka trade name in 60 Oklahoma counties in exchange for \($9,000\) paid to Arrowhead,...
-
Browning Transportation Co. has three regional divisions organized as profit centers. The chief executive officer (CEO) evaluates divisional performance, using income from operations as a percent of...
-
The downward acceleration of a 50-kg stuntwoman near the end of a fall from a very high building is 5.8 m/s2 . What resistive force does the air exert on her body at that point.
-
It is desired to remove some of the n-butane from an equimolar mixture of n-butane and ethane, initially at 25C and 1 bar. The procedure that will be used is to isentropically compress the mixture to...
-
An equimolar mixture of methane and propane at 10C and 1 bar in compressed to 50 bar in a isentropic compressor, cooled back down to 10C, and then undergoes a Joule-Thompson expansion to 10 bar....
-
This chapter described how to create tables and records in Microsoft Access. What other database management systems are available? Use the Internet to learn more about these systems.
-
Choose an Industry 1. Explain the Sentiment Analysis for Product Reviews 2. perform sentiment analysis on a dataset of product reviews and generate insights to help a company understand customer...
-
On January 1, 2021 (Year 1), Chicago Corporation (a calendar year end business) purchased a machine for $35,000 and placed it into service. The machine had an estimated salvage value of $2,500 and a...
-
Using a specific example of contemporary persuasion explain how each of the four eras of media use and its dominant medium would influence how an audience receives, interprets and interacts with that...
-
Ms. Petrov bought her only house for $180,000. In January she sold the house for $500,000, and bought a condo to live in. She paid $30,000 in sales commissions and legal fees on the sale. What would...
-
On January 1, Construction Giants purchased a dump truck for $35,000. It has a salvage value of $5,000 and a useful life of 5 years. What is the book value at the end of the accounting period on...
-
Residents from the Town of Mountain View authorize a $5,000,000 renovation to their historic town hall on November 15, 2013. Financing for the project consists of $2,500,000 from a 5 percent serial...
-
For the following exercises, find the inverse of the function and graph both the function and its inverse. f(x) = 4 x 2 , x 0
-
Which will be more soluble (moles of metal dissolved per liter of solution), Ba(IO 3 ) 2 (K sp = 1.5 10 -9 ) or Ca(IO 3 ) 2 (K sp = 7.1 10 -7 )? Give an example of a chemical reaction that might...
-
Fe(III) precipitates from acidic solution by addition of OH - to form Fe(OH) 3 (s). At what concentration of OH - will [Fe(III)] be reduced to 1.0 10 -10 M? If Fe(II) is used instead, what...
-
Is it possible to precipitate 99.0% of 0.010 M Ce 3+ by adding oxalate (C 2 O 4 2- ) without precipitating 0.010 M Ca 2+ ?
-
Investigate continuity of the function and draw its' graph: 1 f(x)=x-2.5' x <3; /2x-5, x3.
-
7-8. A shop can sell 216 microwaves a week at price of $80. Market researches show that each change of price by $5 changes the quantity of sold TVs for 8 units. At which price microwaves must be sold...
-
The shape of a piece of land is shown. To estimate the area of the land, a surveyor takes measurements at intervals of 50m, perpendicular to the straight portion with the results shown (the...

Study smarter with the SolutionInn App


