Repeat the calculations of Problem 6.13 if the mechanical efficiency of the adiabatic turbine is only 85
Question:
Repeat the calculations of Problem 6.13 if the mechanical efficiency of the adiabatic turbine is only 85 percent.
Problem 6.13
Eighteen kilograms of the refrigerant HFC-134a at 150°C is contained in a 0.03-m3 tank. Compare the prediction you can make for the pressure in the tank with that obtained using Fig. 3.3-4. For data, see Table 6.6-1.
Fig. 3.3-4.
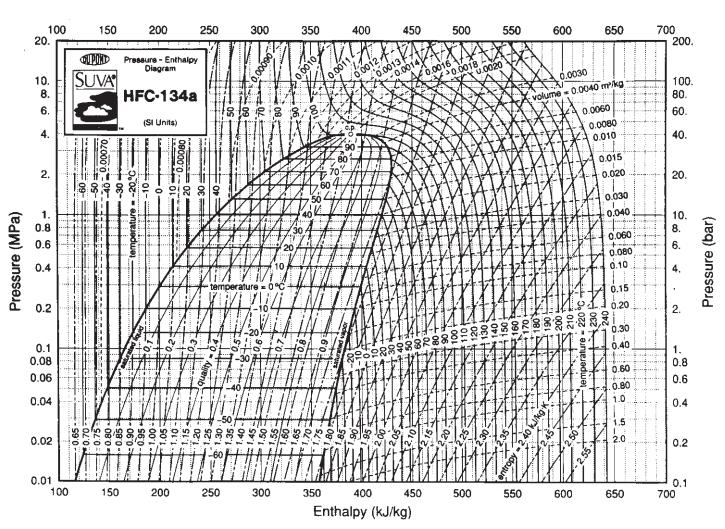
Table 6.6-1.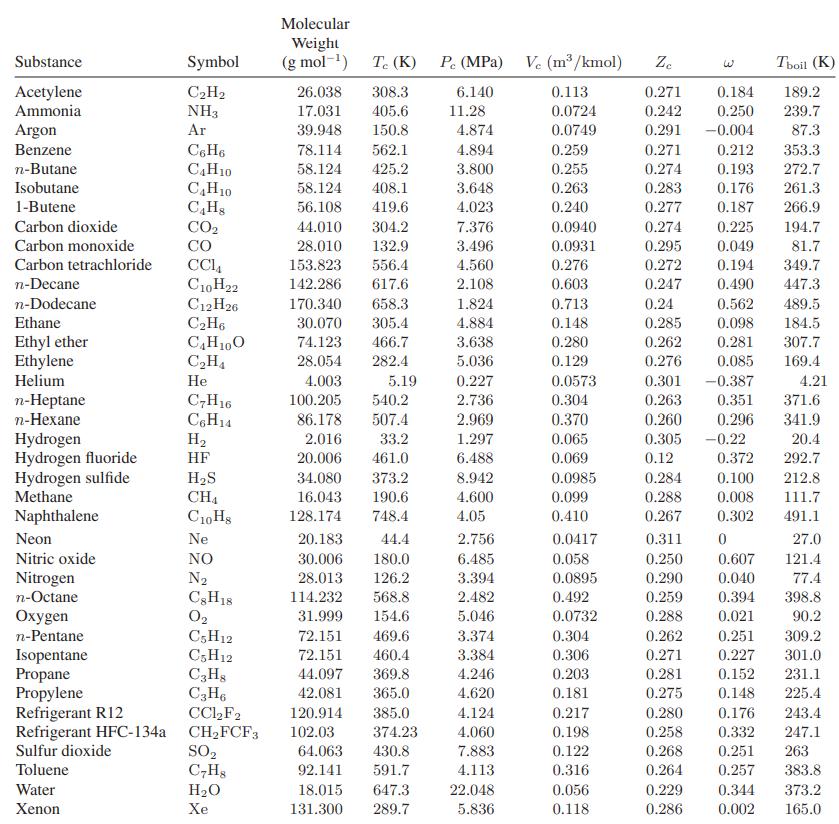
Transcribed Image Text:
Pressure (MPa) 100 20. 10. 8. 6. 4. N 2. 1. 0.8 0.6 0.4 0.2 0.1 0.08 0.06 0.04 0.02 0.01 100 150 QUPORD Pressure - Enthalpy Diagram SUVA HFC-134a (SI Units) 0.65 0.70 0.75 0.00070 -30 temperature -20°C -0.80- -0.85- 0.90- 150 200 960- 250 1.00- 1.05- 1.10- 1.15- 1.20: 200 1885 300 1.35. 1.30 21.25. 250 0.0009 temperature 0°C 1764 20 300 350 0.001 350 400 0.001 0.0013 (0.0012, -0.0014 450 400 450 8888888 Enthalpy (kJ/kg) 0.0016 0.0018 80 500 சம் 100 1104 120+ 500 0.0020 550 FOEK HEREN 140 160 335 41503 NUKITS $65 0/1 EN RECE 550 1801 600 0.0030 volume-0.0040 m²/kg 061 200 temperature 220° BEHI SE 650 0.0060 - 50 600 0.0080 0.010 0.015 0.020 0.030 0.040 0.060 ++ 0.080 0.10 0.15 0.20 0.30 0.40 HIH 0.60 0.80 1.0 O... 1.5 2.0 650 700 200. 100. 80. 60. 40. 8 20. 10. 8. 6. ✔ 2. 1. 0.8 CO CO 0.6 0.4 0.2 0.1 700 Pressure (bar)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
Consider a heat engine and heat pump connected as shown in figure P.7.38. Assume TH1 = TH2 > Tamb and determine for each of the three cases if the setup satisfy the first law and/or violates the 2nd...
-
Redo Problem 6.32 using Aspen Plus Problem 6.32 Repeat the calculations of Problem 6.13 if the mechanical efficiency of the adiabatic turbine is only 85 percent. Problem 6.13 Eighteen kilograms of...
-
Redo Problem 6.13 using Aspen Plus Problem 6.13 Eighteen kilograms of the refrigerant HFC-134a at 150C is contained in a 0.03-m 3 tank. Compare the prediction you can make for the pressure in the...
-
The comparative balance sheets for Karidis Ceramics, Inc., for December 31, 209 and 208 are presented on the next page. During 209, the company had net income of $96,000 and building and equipment...
-
During a period of a few years, intense price competition characterized both the retail and the wholesale oil markets. At times, prices in the wholesale market fell below the manufacturers cost. One...
-
The compressibility factor \(Z\) of an ideal gas is always (a) 0 (b) 1 (c) \(>1\) (d) \( <1\).
-
What are rites of passage and rites of intensification?
-
The following information pertains to the Davenport plant for the month of May (all materials are added at the beginning of the process): Required Compute the cost per equivalent unit for materials...
-
You own a portfolio that isinvested 50 percent in stock A, 15 percent in stock B, and theremainder in stock C. The expected returns on these stocks are14.45 percent, 15.6 percent, and 12.33 percent,...
-
Turtle Electronics Company uses a perpetual inventory system. The unadjusted trial balance for Turtle Electronics Company follows: (Click the icon to view the unadjusted trial balance.) Read the...
-
Nitrogen is to be isothermally compressed at 0C from 1 bar to 100 bar. Compute the work required for this compression; the change in internal energy, enthalpy; Helmholtz and Gibbs energies of the...
-
The speed of propagation of a small pressure pulse or sound wave in a fluid, v S , can be shown to be equal to where is the molar density. a. Show that an alternative expression for the sonic...
-
Those against whom a suit is brought or who are charged with a violation of the law. a. administrative law b. appellate court c. civil law d. common law e. criminal law f. defendants g. express...
-
Landen Corporation uses job-order costing. At the beginning of the year, it made the following estimates: Direct labor-hours required to support estimated production Machine-hours required to support...
-
Rosenberg Sports Inc. manufactures and sells two products, baseball bats and baseball gloves. The fixed costs are $793,600, and the sales mix is 30% bats and 70% gloves. The unit selling price and...
-
Two items are omitted from each of the following three lists of cost of goods sold data from a manufacturing company income statement. Determine the amounts of the missing items, identifying them by...
-
The following data concerning the retail inventory method are taken from the financial records of Crane Company. Cost Retail Beginning inventory $195000 $279000 Purchases 895000 1270000 Freight-in...
-
Explain how and why different communication styles and channels are influenced by various cultural types, such as participative or hierarchical cultures.
-
What factors affect the value of an option?
-
Discrete sample spaces: suppose there are N cable cars in San Francisco, numbered sequentially from 1 to N. You see a cable car at random; it is numbered 203. You wish to estimate N. (See Goodman,...
-
Chemists and engineers must be able to predict the changes in chemical concentration in a reaction. A model used for many single-reactant processes is Rate of change of concentration = -kC n where C...
-
Chemists and engineers must be able to predict the changes in chemical concentration in a reaction. A model used for many singlereactant processes is Rate of change of concentration = -kC n where C...
-
The following list gives the measured gas mileage in miles per gallon for 22 cars of the same model. Plot the absolute frequency histogram and the relative frequency histogram. 23 25 26 25 27 25 24...
-
If following flowchart was executed, what is the final value of variable x? End Start Initialize arrays x=0 k=1 k>3? k=k+1 COMPUTE x=x+k T
-
The issue at hand is whether Burger Queen violated their duty of good faith towards Scheck by opening a new store close to his existing franchise, knowing it would harm his business. Scheck can...
-
Major medical complexes and their service providers continue to move toward advanced health informatics - acquiring, managing, and using information to provide better healthcare. A new analyzer for...

Study smarter with the SolutionInn App


