The following data have been reported for the constant-pressure heat capacity of a benzenecarbon tetrachloride mixture at
Question:
The following data have been reported for the constant-pressure heat capacity of a benzene–carbon tetrachloride mixture at 20°C.
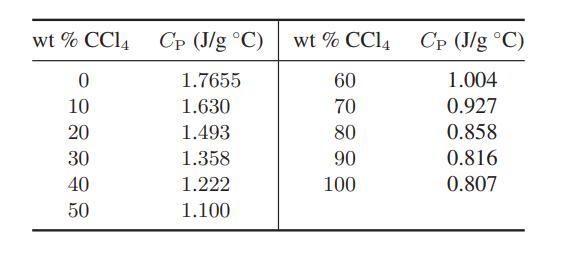
On a single graph plot the constant-pressure partial molar heat capacity for both benzene and carbon tetrachloride as a function of composition.
Transcribed Image Text:
wt% CC14 Cp (J/g °C) 1.7655 1.630 1.493 1.358 1.222 1.100 0 10 20 30 40 50 wt % CC14 60 70 80 90 100 Cp (J/g °C) 1.004 0.927 0.858 0.816 0.807
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
The following data have been reported by Shen and Smith for benzene (Bz) adsorption on silica gel [16]: a) Do these data better fit a single site or a dual site Langmuir isotherm? Why? b) Assuming...
-
The following data have been reported for a sample of 10 major U.S. zoological parks: a. Determine the least-squares multiple regression equation. b. Interpret the y-intercept and partial regression...
-
a. The following data have been reported for the vapor pressure of ethanol as a function of temperature. Use these data to calculate the heat of vaporization of ethanol at 17.33C. b. Ackermann and...
-
You have this information r*=2.5%, inflation premium =3%, default risk premium=3.5%, liquidity premium =2% and maturity risk premium =1.5%.What is the treasury bill rate?
-
Allen, Barker, and Cooper are cosureties on a $750,000 loan by Durham National Bank to Kingston Manufacturing Co., Inc. The maximum liability of the sureties is as follows: Allen, $750,000; Barker,...
-
A defense contractor is interested in studying an inspection process to detect failure or fatigue of transformer parts. Three levels of inspections are used by three randomly chosen inspectors. Five...
-
You need to understand the approach described in question 3 in More Genetic TIPS before answering this question. A gene that is normally expressed in pancreatic cells was cloned and then subjected to...
-
Performance reports and evaluation Vance Corporation has four divisions: the assembly division, the processing division, the machining division, and the packing division. All four divisions are under...
-
A certain substance has a mass per mole of 53 g/mol. When 312 J is added as heat to a 26.0 g sample, the sample's temperature rises from 21.0C to 45.0C. What are the (a) specific heat and (b) molar...
-
The partial molar enthalpies of species in simple binary mixtures can sometimes be approximated by the following expressions: a. For these expressions show that b 1 must equal b 2 . b. Making use of...
-
The temperature achieved when two fluid streams of differing temperature and/or composition are adiabatically mixed is termed the adiabatic mixing temperature. Compute the adiabatic mixing...
-
University of Louisville Professor Julia Karcher conducted an experiment to investigate the ethical behavior of accountants (Journal of Business Ethics, Vol. 15, 1996). She focused on auditor...
-
Linda is a student at Alabama University and does not have any loans. She is just now learning about credit scores and has recently found out that lenders commonly assess the credit payment history...
-
Over the past 25 years, income inequality has been increasing in many developed countries. For example, from 2007 to 2011 Spains Gini coefficient rose by 6.0 percentage points, and Irelands by 6.6...
-
Your task is to create a business proposal for a product or service w hich you believe will work in the Canadian Market. Your Outline will include the following information 1 . Summary of Idea ( The...
-
Why do fascists reject democracy as a form of government? Explain.
-
Implement a function to find the length of the longest common subsequence between two strings using dynamic programming.
-
What is audit risk?
-
You have just begun your summer internship at Omni Instruments. The company supplies sterilized surgical instruments for physicians. To expand sales, Omni is considering paying a commission to its...
-
An automobile assembly plant produces 400 vehicles per day. Make an order-of-magnitude estimate for the weight of the steel needed to make those vehicles. Explain and justify the reasonableness of...
-
Think of some quantity that you encounter in your day-to-day life for which it would be difficult to obtain a highly accurate numerical value but for which an order-of-magnitude approximation can be...
-
The modulus of elasticity, modulus of rigidity, Poissons ratio, and the unit weight for various materials are shown below. The data is given as Material; Modulus of Elasticity, E (Mpsi & GPa);...
-
How do NoSQL databases diverge from traditional relational databases in terms of data modeling paradigms, consistency models, and scalability architectures, and what are the implications for...
-
Suppose the romaine lettuce industry is a Cournot duopoly with the following two firms: Amalgamated Romaine (a) and Best Romaine (b). The (inverse) market demand schedule is: p = 211 -0.5Q...
-
If society is to one day operate sustainably with renewable energies, how might it to be transitioned in such a way that it does not negatively impact individuals or the economy? Do Energy companies,...

Study smarter with the SolutionInn App


