The osmotic pressure for a small molecule in aqueous solution has been found to be given by
Question:
The osmotic pressure for a small molecule in aqueous solution has been found to be given by the following expression
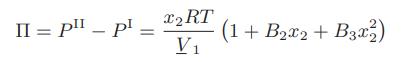
where 1 is the solvent and 2 is the small; molecule solute. Obtain expressions for the activity coefficients of the solvent and solute as a function of mole fraction.
Transcribed Image Text:
II = p _ pl - x2 RT V₁ (1 + B₂x2 + B3x2²2)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The osmotic pressure for a small molecule in aqueous solution can be expressed in terms of activity ...View the full answer

Answered By

AGRAHARAPU SAMPATH KUMAR
I applied to this website so as to do my tutoring job to put full efforts in it and improve my interested subject. I am interested in mathematics and this website has different questions to solve, so I applied to this as a tutor of mathematics.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
A 4 kg/s steady flow of ammonia runs through a device where it goes through a polytropic process. The inlet state is 150 kPa, -20oC and the exit state is 400 kPa, 80oC, where all kinetic and...
-
A 4 kg/s steady flow of ammonia runs through a device where it goes through a polytropic process. The inlet state is 150 kPa, -20oC and the exit state is 400 kPa, 80oC, where all kinetic and...
-
In manufacturing semiconductors, it is necessary to use water with an extremely low mineral content, and to separate water from contaminants, it is common practice to use a membrane process called...
-
The number x of bicycle helmets people are willing to buy per week from a retail chain at a price of $p is given by x = 1,000 - 60p + 25 20 ¤ p ¤ 100 (see the figure). (A) Find dx/dp....
-
In June 1988, British & Commonwealth PLC (B&C) acquired Atlantic Computers, the worlds third largest computer-leasing company. In April 1990, B&C placed Atlantic Computers into administrative...
-
What are the the common (a) objectives and (b) membership requirements of the SEC and Private Companies Practice Sections?
-
True or False. Most phenomena in real life are deterministic.
-
You, CA, are employed at Beaulieu & Beauregard, Chartered Accountants. On November 20, 20X5, Domenic Jones, a partner in your firm, sent you the following email: Our firm has been reappointed...
-
Answer the questions below. (Each 10 marks) 1. Determine the return on investment for the following two video games. Morris Mania Berto's Revenge Manufacturing costs $10.00 $ 8.75 Selling/marketing...
-
A business issued a 60-day, 9% note for $61,000 to a creditor on account. Illustrate the effects on the accounts and financial statements of recording (a) the issuance of the note and (b) the payment...
-
The more environmentally friendly refrigerants are made of hydrogen-containing compounds that have a short environmental lifetime. However, these compounds are also combustible. Therefore, one...
-
A new membrane premeable only to water is to be used for purifying water containing a pesticide. Your employer requests that you setup a demonstration consisting of an insulated rigid tank at 2C...
-
What is the purpose of an unadjusted trial balance?
-
Microsoft has a bond that was issued two years ago. It has a par value of $3,000, the coupon rate of 9%, paid semiannually , and has 4 more years left until it matures. Its yield to maturity is 10%....
-
Assume a world without inflation. Janice Lewis berg, 38, plans to retire at age 68. Her life expectancy is age 90. She wants to live a retirement lifestyle that will cost $35,000 per year, payable at...
-
Mark's yearly salary is $89,000 and he deposits 25% of it into his savings account at the end of every year. The interest rate the bank offers is 3.1% per year. It has been 3 years since Mark started...
-
How do operating systems implement security measures for inter-process communication (IPC), and what are the potential security risks associated with IPC mechanisms like shared memory and message...
-
Evaluate the concept of endpoint detection and response (EDR) in the context of operating system security. What are the key components of an EDR solution, and how does it differ from traditional...
-
An FI has purchased a $ 200 million cap of 9 percent at a premium of 0.65 percent of face value. A $ 200 million floor of 4 percent is also available at a premium of 0.69 percent of face value. a. If...
-
The water in tank A is at 270 F with quality of 10% and mass 1 lbm. It is connected to a piston/cylinder holding constant pressure of 40 psia initially with 1 lbm water at 700 F. The valve is opened,...
-
Write the names and abbreviations for each of the prefixes from 10 -24 to 10 24 . Which abbreviations are capitalized?
-
What is the formal concentration (expressed as mol/L = M) of NaCl when 32.0 g are dissolved in water and diluted to 0.500 L?
-
How many grams of boric acid, B(OH) 3 (FM 61.83), should be used to make 2.00 L of 0.050 0 M solution? What kind of flask is used to prepare this solution?
-
A parallel-plate capacitor is to be constructed by using, as a dielectric, rubber with a dielectric constant of 3.20 and a dielectric strength of 15.0 MV/m. The capacitor is to have a capacitance of...
-
1. A car is traveling at 60 mph and needs to come to a stop in 100 feet. What acceleration is required if the brakes are applied after a 0.5-second delay? 2. A ball is rolled around a circular track...
-
A 7400 kg rocket blasts off vertically from the launch pad with a constant upward acceleration of 2.20 m/s2 and feels no appreciable air resistance. When it has reached a height of 600 m , its...

Study smarter with the SolutionInn App


