Here are data from a (mathrm{pH}) titration of potassium hydrogen phthalate with (mathrm{NaOH}) using an automated titrator.
Question:
Here are data from a \(\mathrm{pH}\) titration of potassium hydrogen phthalate with \(\mathrm{NaOH}\) using an automated titrator. Only data for part of the titration has been provided. Prepare a Gran plot and find the equivalence point volume.
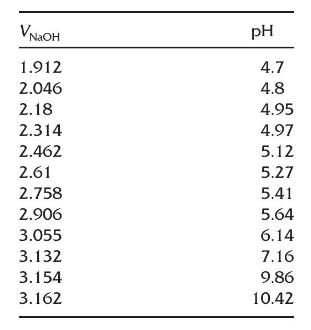
Transcribed Image Text:
NGOH 1.912 pH 4.7 4.8 2.046 2.18 4.95 2.314 4.97 2.462 5.12 2.61 5.27 2.758 5.41 2.906 5.64 3.055 6.14 3.132 7.16 3.154 9.86 3.162 10.42
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
VNaoH pH VNaoH10PH 0000045 1912 47 38149E05 2046 218 48 32427E05 495 2446E05 2314 2462 49...View the full answer

Answered By

Mario Alvarez
I teach Statistics and Probability for students of my university ( Univerisity Centroamerican Jose Simeon Canas) in my free time and when students ask for me, I prepare and teach students that are in courses of Statistics and Probability. Also I teach students of the University Francisco Gavidia and Universidad of El Salvador that need help in some topics about Statistics, Probability, Math, Calculus. I love teaching Statistics and Probability! Why me?
** I have experience in Statistics and Probability topics for middle school, high school and university.
** I always want to share my knowledge with my students and have a great relationship with them.
** I have experience working with students online.
** I am very patient with my students and highly committed with them
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Electroanalytical Chemistry Principles Best Practices And Case Studies
ISBN: 9781119538592,9781119538585
1st Edition
Authors: Gary A. Mabbott
Question Posted:
Students also viewed these Engineering questions
-
A solution of NaOH was standardized by titration of a known quantity of the primary standard, potassium hydrogen phthalate The NaOH was then used to find the concentration of an unknown solution of H...
-
A solution of NaOH was standardized by gravimetric titration of a known quantity of the primary standard, potassium hydrogen phthalate: Potassium hydrogen phthalate C 8 H 5 O 4 K, FM 204.22 The NaOH...
-
Potassium hydrogen phthalate is a primary standard used to measure the concentration of NaOH solutions. Find the true mass of potassium hydrogen phthalate (density = 1.636 g/mL) if the mass weighed...
-
Assume that the average talk time on an Apple iPhone is 20 hours and that this time follows the exponential probability distribution. What is the probability that a randomly selected iPhone will...
-
Tyler and Stephanie work for a direct marketing firm. They make calls to customers for a local carpet cleaning service. In a typical hour, Tyler completes 50 calls and gets two sales Stephanie...
-
What do you think of the Nielsen Holdings CEOs decision to appoint himself, a white male, chief D&I officer? Consider pros and cons, plus how circumstances might make a difference in appraising its...
-
A university pumps its water from wells located on campus. The falling water table has caused pumping costs to increase, the quantity of water available to decrease, and the quality of water to...
-
The accountant for Teresa's Dress Shop prepared the following cash budget. Teresa's desires to maintain a cash cushion of $14,000 at the end of each month. Funds are assumed to be borrowed and repaid...
-
4. Consider a quantum system Q described by a Hilbert space H. (a) Suppose we are given a subspace Ho of H and a linear map from kets in Ho to others in h. That is, Vo)), in a linear way. This map...
-
Why is the filling port for the outer reference chamber of a combination glass electrode kept open during measurements?
-
Imagine that you were asked to set up a flow injection system in order to measure \(\mathrm{K}^{+}\)ion levels in blood samples in a hospital lab. List at least five different challenges for...
-
Let C2 have the inner product of Exercise 1. Lind ||w|| if (a) w = (- i, 3i) (b) w = (0, 2 - i)
-
What is ethical blind spot in forensic psychology? What is the blind spot in human behavior?
-
What are 3 educational programs and ways to gain the experience of clinical health psychology, community health psychology, occupational health psychology, and public health psychology?
-
Define social psychology and explain how social psychology is an experimental branch of the discipline of psychology in general ?
-
Alexis Texas, the CFO of Mullet Production Company, is planning next year's capital budget. It is at its optimal capital structure, which is 40 percent debt and 60 percent common equity, and the...
-
What is the difference between structuralism and functionalism in psychology?
-
Water flows through a Venturi meter, as shown in Fig.P1.36. The pressure of the water in the pipe supports columns of water that differ in height by 10. Determine the difference between points a and...
-
Use the information given about the angles and to find the exact value of: (a) sin( + ) (b) cos( + ) (c) sin( - ) (d) tan ( + ) (e) sin(2) (f) cos (2) (g) sin /2 (h) cos/2 cos = 4/5, 0 < < /2; cos =...
-
What will be change in the heat transfer coefficient in problem 2 after the modification? Data from in problem 2 In a rising film evaporator, if the heat transfer coefficient is proportional V0.6,...
-
In a rising film evaporator, if the heat transfer coefficient is proportional V0.6, where V is the rising vapor velocity. If the throughput through the reactor has to be increased by 25%, how many...
-
How long it will take to reduce the pressure of a 100 L tank from 1 Kg/cm2 (a) to 0.2 Kg/cm2 (a), if the pump capacity is 500 L/min. assume 10% leakage.
-
The "war on drugs" has had a very real and significant impact on both communities and the corrections system. How has the "war on drugs" consumed valuable resources and what impact has it had on the...
-
Discuss how can people identify what they are really good at and pursue careers related to their natural abilities.
-
Provide an overview of Mark 12:13-17. What does the Bible say about taxation Is there a level of taxation that is sinful or too much Explain fully. What is the optimal level of taxation for...

Study smarter with the SolutionInn App


