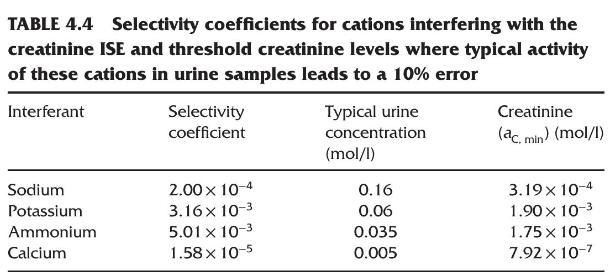
Refer to Table 4.4 of selectivity coefficients for cations that interfere with the creatinium ISE described. At
Question:
Refer to Table 4.4 of selectivity coefficients for cations that interfere with the creatinium ISE described. At what concentration of creatinine would \(0.3 \mathrm{M}\) ammonium interfere to cause a \(10 \%\) error in the measured creatinine concentration?
Transcribed Image Text:
TABLE 4.4 Selectivity coefficients for cations interfering with the creatinine ISE and threshold creatinine levels where typical activity of these cations in urine samples leads to a 10% error Interferant Selectivity coefficient Typical urine Creatinine concentration (ac, min) (mol/l) (mol/l) Sodium 2.00 x 10-4 0.16 3.19 10-4 Potassium 3.16 x 10-3 0.06 1.90 x 10-3 Ammonium 5.01 x 10-3 0.035 1.75 x 10-3 Calcium 1.58 x 10-5 0.005 7.92 x 10-7
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
RT Ememb E 1 F ln ax KKNH AN...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Electroanalytical Chemistry Principles Best Practices And Case Studies
ISBN: 9781119538592,9781119538585
1st Edition
Authors: Gary A. Mabbott
Question Posted:
Students also viewed these Engineering questions
-
Refer to Table 4.4 for the selectivity coefficients for ions that interfere with the determination of creatinine by the experimental ISE described in Section 4.3. Assume that \(\mathrm{Na}^{+},...
-
Refer to Table 4 in the chapter, which assumes the country continues to pay an interest rate of 8% on its debt. Suppose, instead, that the interest rate were 10%, while each years nominal GDP and...
-
The Trial Balance and Adjustments columns of the worksheet of Wells Decorating Centre included these accounts and balances at December 31, 2017: Required Wells Decorating Centre uses the perpetual...
-
The CEO of Jump Up and Down Workout Equipment Company remarked to a colleague, "I don't understand why other companies waste so much time and effort in the budgeting process. I set our company goals,...
-
Why is it so hard to make decisions that weight longterm more than short-term considerations? What examples, personal and in current events, can you think of?
-
Consider the following cash flow profile and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically attractive?...
-
Disclosures Required in Various Situations Ace Inc. produces electronic components for sale to manufacturers of radios, television sets, and digital sound systems. In connection with her examination...
-
5. Suppose that acts on density operators for Q with Kraus representation () - k=1 Let U M, (C), and define operators = 1, k BkUkjAj. , Show that B,..., B., are also Kraus operators for E. 6. Even...
-
For a membrane without an ionophore, why is the membrane potential more sensitive to chlorate ion than sulfate ion?
-
A well-used nitrate ISE was being used to test some ground water samples for possible leaching from a hazardous waste site. A percent recovery procedure was performed to decide whether a standard...
-
Consider a dynamic system with the following transfer function, \[G(s)=\frac{s+2}{(s+3)(s+4)}\] Draw the Bode plots of the system.
-
Explain who was Charlemagne? Discuss several of his accomplishments. In a separate paragraph, separated by a blank line...Discuss what you think was his most important accomplishment and why you...
-
There are various known branches of psychology. List these major branches of psychology.?
-
Answer to the following: Discuss how a company's internal environment might affect the development of the corporate strategy. Include product life cycle, personnel, and organizational structure in...
-
1)What makes psychology a science? 2) Do you agree with Stanovich that psychology is "alive and well" among the sciences? Why or why not? Stanovich: how to think straight about psychology
-
What are some examples of how professionals in the field of psychology help individuals maximize memory?
-
As shown in fig 1.7, a manometer is attached to a tank of gas in which the pressure is 104.0 kPa. The manometer liquid is mercury, with a density of 13.59 g/cm3. If g = 9.81 m/s2 and the atmospheric...
-
A new car sold for $31,000. If the vehicle loses 15% of its value each year, how much will it be worth after 10 years?
-
What is the heat required to evaporate 1000 kgs of water at 100C?
-
Estimate the Resolution between two peaks A and B if the time at which peak maximum occurs are 8 and 12 min, respectively. The widths at the base are 3 and 4 min, respectively.
-
Derive the mathematical equation relating the width at the base and width at half height in a chromatogram
-
Six grocery items have the following prices: $5.82, $10.28, $8.51, $12.13, $6.64, and $11.77. (a) Estimate the total cost of the six items by rounding each price to the nearest dollar, then adding....
-
KBC, Inc. has the financial profile illustrated below. Next year, Margins (%) will hold steady, and Assets grow proportionally with Sales. Debt will remain unchanged. ABC intends to maintain the same...
-
SALEM Corporation Financial Analysis Background: SALEM Corporation is a manufacturing company that produces and sells electronic gadgets. The company has been in operation for the past five years and...

Study smarter with the SolutionInn App


