A multicomponent mixture is boiled in a flask at (1 mathrm{~atm}). The vapors are condensed and recovered
Question:
A multicomponent mixture is boiled in a flask at \(1 \mathrm{~atm}\). The vapors are condensed and recovered as a liquid product. It is desired to examine the mole fractions of the residual liquid in the flask as vaporization proceeds. Although sketches of the residue-curve maps are called for in (b)-(d), a process simulator can be used to prepare the drawings accurately.
(a) For a mixture of \(60 \mathrm{~mol} \% \mathrm{n}\)-butane (1) and \(40 \mathrm{~mol} \% \mathrm{n}\)-pentane (2), determine the residual mole fraction of \(n\)-butane after \(10 \%\) of the liquid has vaporized.
(b) Consider mixtures of n-butane (1), n-pentane (2), and n-hexane (3). For three typical feed compositions:
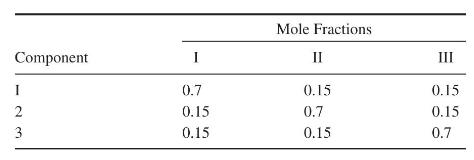
Sketch the residue curves (solutions of the ODEs; do not solve them analytically or numerically) on triangular graph paper. Use arrows to show the direction along the trajectories in time.
(c) Repeat
(b) for mixtures of acetone (1), choroform (2), and benzene (3). Note that the acetone-chloroform binary exhibits a maximum-boiling azeotrope \(\left(64^{\circ} \mathrm{C}\right)\) at \(35 \mathrm{~mol} \%\) acetone with no other azeotropes existing. Sketch any boundaries across which the residue curves cannot traverse.
(d) Repeat
(c) for mixtures of methyl acetate (1), methanol (2), and \(\mathrm{n}\)-hexane (3). Note the existence of four azeotropes, where compositions are in \(\mathrm{mol} \%\).
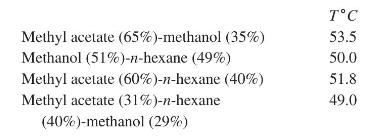
Step by Step Answer:

Product And Process Design Principles Synthesis Analysis And Evaluation
ISBN: 9781119355243
4th Edition
Authors: Warren D. Seider, Daniel R. Lewin, J. D. Seader, Soemantri Widagdo, Rafiqul Gani, Ka Ming Ng




