Calculate the standard heat of the following reaction, given the enthalpies of formation: Standard enthalpies of formation
Question:
Calculate the standard heat of the following reaction, given the enthalpies of formation:
![]()
Standard enthalpies of formation kJ/mol
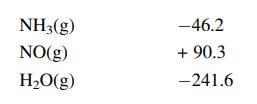
Transcribed Image Text:
4NH3(g) +50₂(g) →4NO(g) + 6H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The enthalpy of formation ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Engineering Design
ISBN: 9780081025994
6th Edition
Authors: Ray Sinnott, R.K. Sinnott, Sinnott Gavin Towler
Question Posted:
Students also viewed these Engineering questions
-
You have been given the task of determining the standard heat of the reaction in which calcium chloride hexahydrate is formed from anhydrous calcium chloride: CaCl 2 (s) + 6 H 2 O (l) CaCl 2 6H 2...
-
Calcium chloride is a salt used in a number of food and medicinal applications and in brine for refrigeration systems. Its most distinctive property is its affinity for water: in its anhydrous form...
-
Trichloroethylene, a widely used degreasing solvent for machine parts, is produced in a two-step reaction sequence. Ethylene is first chlorinated to yield tetrachloroethane, which is...
-
1-Is it a good business strategy to have a day care center, Harley Davidson Motorcycle Dealership Sales and repair, Namaste Yoga studio, bookstore, temporary worker (day laborer) center, state prison...
-
The following paragraphs appeared in the New York Times on September 22, 1986. To keep the dollar from falling against the West German mark, the European central banks would have to sell marks and...
-
Your goal is to solve the following programming lab activity. Write a program that prompts and accepts a number between 1 and 12. After getting the input, display the number with the appropriate...
-
What forms of negligence are described in this chapter?
-
Discuss how a nations market size might impact international trade and, in turn, international logistics.
-
(QUESTIONS) 1. Facts of the Case Provide a very concise and clear summary of the case. Be brief but do include all information that you feel is essential to the case. 2. Arguments provided by the...
-
Hydrogen chloride gas, produced by burning chlorine with hydrogen, is required at a supply pressure of 600 kN/m 2 , gauge. The pressure can be achieved by either operating the burner under pressure...
-
Hydrogen chloride is produced by burning chlorine with an excess of hydrogen. The reaction is highly exothermic and reaches equilibrium very rapidly. The equilibrium mixture contains approximately 4...
-
In a study of the cognitive capacities of non-human primates, 19 monkeys of the same age are randomly divided into two groups of 10 and 9. The groups are trained by two different teaching methods to...
-
Candice's' Cupcakes is a new cupcake delivery company in Gainesville, Florida. The firm hires local college students to sell cupcakes door-to-door. Each of these "Sales Associates" sells cupcakes,...
-
A group of researchers collected information about labor force participation and sex among 11,920 individuals and created the following cross-tabulation with frequencies. What is the P(Male | No...
-
1. Exercise 30.1 (Variants of the Stag Hunt) Consider variants of the n-hunter Stag Hunt. That is, there are n number of hunters. Only m hunters, with 2 m
-
The top player on the Crabs is Hanson Edelmann and they eam $189,500 per season. Assuming this is their entire income answer the following questions using the table above: What is their total income...
-
What is the gauge pressure of the air inside an alveolus having a radius of 250 m if the surface tension of the fluid-lined wall is the same as for soapy water (37), and the gauge pressure in the...
-
For the following functions of the accounting system, identify technologies used by each function. 1. Storing accounting data 2. Analyzing accounting data 3. Safeguarding accounting data
-
Respond to the ethical judgments required based on the following scenarios. Scenario 1. Assume you have collected a sample using MUS and that you have evaluated that sample to calculate a total...
-
Suggest suitable materials of construction for the following applications: 1. A 10,000 m 3 storage tank for toluene; 2. A 5:0 m 3 tank for storing a 30% w/w aqueous solution of sodium chloride; 3. A...
-
Choose a suitable material of construction for the following duties: 1. 98% w/w sulfuric acid at 708C; 2. 5% w/w sulfuric acid at 308C; 3. 30% w/w hydrochloric acid at 508C; 4. 5% aqueous sodium...
-
A pipeline constructed of carbon steel failed after 3 years of operation. On examination it was found that the wall thickness had been reduced by corrosion to about half the original value. The...
-
Everett Company's inventory at December 31 and the costs charged to Work in Process-Department B during December are as follows: 500 units, 60% completed From Department A, 10,000 units Direct labor...
-
Wesley Company makes bowling balls and uses the total cost method in setting product prices. Its costs for producing 10,000 bowling balls follow. The company targets a 12.5% markup on total cost. The...
-
A customer is angry. He/She does not like a service provided by a company. He/She claims his/her money back. He/she want to get 100% of this sum. According to a contract a customer signed with a...

Study smarter with the SolutionInn App


