Estimate the critical constants for diphenylmethane using Lydersens method; normal boiling point 537.5 K, molecular mass 168.2,
Question:
Estimate the critical constants for diphenylmethane using Lydersen’s method; normal boiling point 537.5 K, molecular mass 168.2, structural formula:
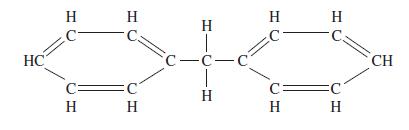
Transcribed Image Text:
HC H С H Н C. C Н H T c-С-С | H Н Н H C H CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Group HCring Cring CHz No of 10 2 1 Total contr...View the full answer

Answered By

Antony Sang
I am a research and academic writer whose work is outstanding. I always have my customer's interests at heart. Time is an important factor in our day to day life so I am always time conscious. Plagiarism has never been my thing whatsoever. I give best Research Papers, Computer science and IT papers, Lab reports, Law, programming, Term papers, English and literature, History, Math, Accounting, Business Studies, Finance, Economics, Business Management, Chemistry, Biology, Physics, Anthropology, Sociology, Psychology, Nutrition, Creative Writing, Health Care, Nursing, and Articles.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Engineering Design
ISBN: 9780081025994
6th Edition
Authors: Ray Sinnott, R.K. Sinnott, Sinnott Gavin Towler
Question Posted:
Students also viewed these Engineering questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
a. Using the relationships derived in Example Problem 7.1 and the values of the critical constants for water from Table 7.2, calculate values for the van der Waals parameters a, b, and R from z c , T...
-
The following table shows the five scores from an Introductory Statistics course. Find a model for final sure to check the conditions for multiple regressions. 2 5 6 3 5 7 2 5 3 10 1 1 9 1 8 8 9 9 0...
-
Problems 113 122. The purpose of these problems is to keep the material fresh in your mind so that you are better prepared for later sections, a final exam, or subsequent courses such as calculus. X...
-
Name three methods of cost estimation.
-
Now is the time to begin evaluating your project, measuring it against marketing goals. Ask yourself the following questions. How is your design supporting or extending the brand experience? How is...
-
In 1970, Rose Mary Knick purchased 90 acres of land in Scott Township, Lackawanna County, Pennsylvania. In 2008, another resident of Scott Township discovered documents that suggested that one of...
-
Refer to the problem chart for the automobile oil change in solved problem 2. Calculate the annual labor cost if: The mechanic earns $40 per hour (including variable fringe benefits). The process is...
-
An electric power company regulated by the state is a monopoly enterprise. 1Suppose the regulator sets the price to reflect what the market will look like if it is competitive. 2Now suppose that the...
-
A precipitate is to be continuously separated from a slurry. The solids concentration is 5 per cent and the slurry feed rate 5.5 m 3 /h. The relevant physical properties at the system operating...
-
Estimate the surface tension of pure methanol at 20 C, density 791.7 kg/m 3 , molecular weight 32.04.
-
In Problem, use implicit differentiation to find y and evaluate y at the indicated point. 3xy - 2x - 2 = 0; (2,1)
-
A random wizard deposits $2,718.28 in an account earning 1.82% interest compounded 8 times per year. The wizard withdraws $1,000 after 4 ycars and another $1,000 after 5 years. How much can be...
-
With non-mutually exclusive projects. a. the payback method will select the best project. b. the net present value is not acceptable. c. the internal rate of return method will always select the best...
-
You have bought a bond which carries a coupon rate of 8 percent, has 7 years until maturity, and sells at a yield to maturity of 7 percent. Show your calculations and answer the following questions...
-
Write a java program that reads and saves 10 values from the user in an array. Then calls function smallest Distance The smallestDistance function will finds two neighboring numbers in an array with...
-
Assume the following information for five different types of bonds: Face value at maturity ($) Term to maturity (years) Coupon rate (coupon paid annually) (%) Current bond price ($) Callable after 3...
-
Dust Busters Inc. manufactures two types of small hand-operated vacuum cleaners. Dust Busters is concerned about quality issues and has compiled the following information for the past year associated...
-
Starr Co. had sales revenue of $540,000 in 2014. Other items recorded during the year were: Cost of goods sold ..................................................... $330,000 Salaries and wages...
-
Calculate the cathode potential (versus S.C.E.) needed to reduce cobalt(II) to 1.0 M in each of the following solutions. In each case, Co(s) is the product of the reaction. (a) 0.10 M HClO 4 (b) 0.10...
-
Ions that react with Ag + can be determined electrogravimetrically by deposition on a silver working anode: (a) What will be the final mass of a silver anode used to electrolyze 75.00 mL of 0.023 80...
-
Chlorine has been used for decades to disinfect drinking water. An undesirable side effect of this treatment is reaction with organic impurities to create organochlorine compounds, some of which...
-
Tina, an estate planning client of yours would like her husband to benefit from her property for his lifetime if he survives her. However, Tina is adamant that her husband not benefit from her...
-
the following data ( Use data below since data is not same as textbook ) detergentA = ( 8 4 , 8 3 , 8 7 , 8 5 , 9 4 , 8 9 , 8 9 , 8 3 ) detergentB = ( 7 8 , 8 0 , 8 1 , 8 6 , 8 6 , 8 9 , 6 9 , 7 9 )...
-
solve 85-8 P(X = 5) = 10.99

Study smarter with the SolutionInn App


