(a) From data in Appendix 2A, derive a numerical form of the ClausiusClapeyron equation for methanol. (b)...
Question:
(a) From data in Appendix 2A, derive a numerical form of the Clausius–Clapeyron equation for methanol.
(b) Use the equation to plot the appropriate quantities that should give a straightline relation between vapor pressure and temperature.
(c) Estimate the vapor pressure of methanol at 0.0°C.
(d) Estimate the normal boiling point of methanol.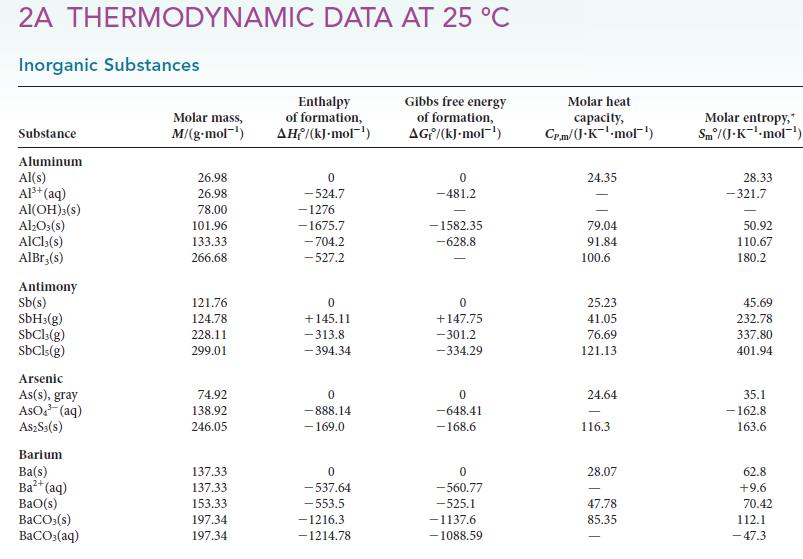
Transcribed Image Text:
2A THERMODYNAMIC DATA AT 25 °C Inorganic Substances Substance Aluminum Al(s) Al³+ (aq) Al(OH)3(S) Al₂O3(s) AlCl3(s) AlBr,(s) Antimony Sb(s) SbH3(g) SbCl3(g) SbCls (g) Arsenic As(s), gray AsO³(aq) A$2S3(S) Barium Ba(s) Ba²+ (aq) BaO(s) BaCO3(s) BaCO3(aq) Molar mass, M/(g.mol-¹) 26.98 26.98 78.00 101.96 133.33 266.68 121.76 124.78 228.11 299.01 74.92 138.92 246.05 137.33 137.33 153.33 197.34 197.34 Enthalpy of formation, AH/(kJ-mol-¹) 0 -524.7 -1276 -1675.7 -704.2 -527.2 0 +145.11 -313.8 -394.34 0 -888.14 - 169.0 0 -537.64 -553.5 -1216.3 -1214.78 Gibbs free energy of formation, AG/(kJ.mol-¹) 0 -481.2 -1582.35 -628.8 0 +147.75 -301.2 -334.29 0 -648.41 -168.6 0 -560.77 -525.1 -1137.6 -1088.59 Molar heat capacity, Cr.m/(J.K¹-mol¹) 24.35 79.04 91.84 100.6 25.23 41.05 76.69 121.13 24.64 116.3 28.07 47.78 85.35 Molar entropy, Sm/(J-K-¹-mol¹) 28.33 -321.7 50.92 110.67 180.2 45.69 232.78 337.80 401.94 35.1 -162.8 163.6 62.8 +9.6 70.42 112.1 -47.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a b The relationship ...View the full answer

Answered By

Seema kuldeep
although I don't have an experience of teaching in a particular institute, previously I was an expert on Chegg and I have used to teach my batch mates and also my juniors.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
(a) From data in Appendix 2A and Table 4C.1, derive a numerical form of the ClausiusClapeyron equation for benzene. (b) Use the equation to plot the appropriate quantities that should give a...
-
In the process of distillation, a mixture of two (or more) volatile liquids is first heated to convert the volatile materials to the vapor state. Then the vapor is condensed, reforming the liquid....
-
The stock market consists of only 2 stocks, X and Y Their market capitalizations are $1.5bn and $8.5bn respectively. Their returns are uncorrelated. The mean return of X is 6% and that of Y is 11%....
-
Suppose that two linear equations are graphed on the same set of coordinate axes. Sketch what the graph might look like if the system has the given description. (a) The system has a single solution....
-
Staying up late to study, and having no stove to heat water, you use a 200-W heater from the lab to make coffee throughout the night. If 90% of the energy produced by the heater goes toward heating...
-
What is a debt guarantee and how does the SBA back a small business loan?
-
Use the equation (17.19) suggested by Pai for the turbulent stress and integrate for the velocity profile. How do the results compare with that of Prandtl? = 0.9835 H+ (1-#)['-(1-#)'] (17.19) H+
-
The 2010 and 2009 comparative balance sheets and 2010 income statement of King Supply Corp. follow: King Supply had no noncash investing and financing transactions during 2010. During the year, there...
-
2. Find the worst-case time complexity of the OpenMystery algorithm below. Input: n: positive integer 1 Algorithm: OpenMystery 2 Let open be a Boolean array of size n 3 Initialize open to false 4...
-
There are only two risky assets (stocks) A and B in the market. Mean 20% B 10% The returns on the two assets have zero correlation. A Standard Deviation 10% 5% A. Assume that there is no risk-free...
-
A polymer sample of mass 0.20 g dissolved in 0.100 L of toluene, gives rise to an osmotic pressure of 6.3 Torr at 20C. What is the molar mass of the polymer?
-
The normal boiling point of ethyl acetate, CH 3 COOC 2 H 5 , used to remove nail polish, is 77.1C, and its vapor pressure at 16.2 C is 10.0 kPa. Calculate (a) The standard enthalpy of vaporization of...
-
Hitachi, Ltd., reports total revenues of 9,041,071 million for its fiscal year ending March 31, 2013, and its March 31, 2013, unadjusted trial balance reports a debit balance for trade receivables...
-
What are the four advantages of private equity funds? How do they help alleviate the free-rider problem?
-
What are the advantages and disadvantages of using forward contracts to hedge?
-
Why might the market be the best mechanism for minimizing conflicts of interest?
-
How can financial derivatives create excessive risk in the financial system?
-
It is July 30, 2021. The cheapest-to-deliver bond in a September 2021 Treasury bond futures contract is a 13% coupon bond, and delivery is expected to be made on September 30, 2021. Coupon payments...
-
How much interest would be charged on a line of credit that charges a monthly interest rate of 0.75% if the average daily balance for the month were $26,200?
-
Software Solution is family-owned business that has been in operation for more than 15 year. The board of directors is comprised of mainly family members, plus a few professionals such as an...
-
Propose a plausible mechanism for each of the following reactions: a. b. Br Br2 . [H,SO,]
-
Propose a plausible mechanism for the following process, called iodolactonization: I2
-
When 3-bromocyclopentene is treated with HBr, the observed product is a racemic mixture of trans-1,2-dibromocyclopentane. None of the corresponding cis-dibromide is observed. Propose a mechanism that...
-
Houston - based Advanced Electronics manufactures audio speakers for desktop computers. The following data relate to the period just ended when the company produced and sold 4 1 , 0 0 0 speaker sets:...
-
Lopez Sales Company had the following balances in its accounts on January 1 , Year 2 : Cash $ 6 2 , 0 0 0 Merchandise Inventory 4 2 , 0 0 0 Land 1 0 2 , 0 0 0 Common Stock 8 2 , 0 0 0 Retained...
-
Why do we sometimes record a revenue or expense as a deferred item? Wouldn t it be easier to always record revenue to a revenue account and remove any unearned revenue at year - end?

Study smarter with the SolutionInn App


