At what temperatures is the following process spontaneous at 1 atm? What is the normal boiling point
Question:
At what temperatures is the following process spontaneous at 1 atm?
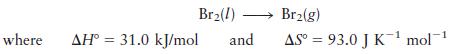
What is the normal boiling point of liquid Br2?
Transcribed Image Text:
Br(1) Br(g) where AH 31.0 kJ/mol and AS= 93.0 J K mol-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The vaporization process will be spontaneous at all temperatures at which AG is negative Note that A...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Sunshine Service Center received a 120-day, 6% note for $40,000 dated April 12 from a customer on account. Assume a 360 day year. a) Determine the due date of the note?? b) Determine the maturity...
-
Like most substances, bromine exists in one of the three typical phases. Br2 has a normal melting point of 27.2oC and a normal boiling point of 59oC. The triple point for Br2 is 27.3oC and 40 torr,...
-
Liquid nitrogen is stored in 0.5-m3 metal tanks that are thoroughly insulated. Consider the process of filling an evacuated tank, initially at 295 K. It is attached to a line containing liquid...
-
In the United States, a principal responsibility for preserving endangered species (e.g., a pair of endangered birds that chooses to nest on private land) and the costs of exercising that...
-
A clown is juggling four balls simultaneously. Students use a video tape to determine that it takes the clown 0.9 s to cycle each ball through his hands (including catching, transferring, and...
-
A rectangular gate is installed in a vertical wall of a reservoir, as shown in Fig. 4.26. Compute the magnitude of the resultant force on the gate and the location of the center of pressure. Also...
-
Mary Kay Morrow began working for Hallmark in 1982. At the beginning of 2002, Hallmark adopted the Hallmark Dispute Resolution Program, which required, among other things, that claims against the...
-
The following gives the number of accidents that occurred on Florida State Highway 101 during the past 4 months: Month Number of Accidents January ........... 30 February.......... 40 March...
-
13. An analyst is valuing Red Inc. common stock using the dividend discount model. The company plans to start paying dividends with its first dividend of $3.25 per share occurring next year. To...
-
Predict the sign of S for each of the following reactions. a. the thermal decomposition of solid calcium carbonate: b. the oxidation of SO 2 in air: CaCO3(s) CaO(s) + CO,(g)
-
In the metallurgy of antimony, the pure metal is recovered by different reactions, depending on the composition of the ore. For example, iron is used to reduce antimony in sulfide ores: Carbon is...
-
In problem proceed as in Example 3 to find a solution of the given initial-value problem. Evaluate the integral that defines y p (x). y'' + y = sec 2 x, y() = 0, y'() = 0
-
Where is the Caldwells' cancellation of debt income first entered on their tax return?
-
he precision of the expectation Blank______. Multiple select question. needs to be very precise if the assertion being tested requires a high level of detection risk is a function of materiality and...
-
Yow will write an original anecdote this story titled "little match girl. his can be a continuation of your introduction or a moral lesson that you learned from the story. ...
-
Ali is an investor with a high-risk tolerance whose primary investment objective is capital gains. He is a high-income earner, so he does not want to receive current income from his investments....
-
What was one of the disturbing trends noted by one of the technical managers, and why what this a problem? (15 points) How did the technical managers at BCC use systems thinking to address the...
-
Kim & Smith Refiners starts business on March 1. The following operations data are available for March for the one lubricant it produces: Gallons Beginning inventory . . . . . . . . . . . . . . . . ....
-
Federated Shipping, a competing overnight delivery service, informs the customer in Problem 65 that they would ship the 5-pound package for $29.95 and the 20-pound package for $59.20. (A) If...
-
Identify the number of valence electrons (including d-electrons) present in each of the following metal ions: (a) Co 2+ ; (b) Mo 4+ ; (c) Ru 4+ ; (d) Pt 2+ ; (e) Os 3+ ; (f) V 3+ .
-
The complex ion [Ni(NH 3 ) 6 ] 2+ forms in a solution containing 0.16 mol L 1 NH 3 (aq) and 0.015 mol L 1 Ni 1 (aq). If the formation constant of [Ni(NH 3 ) 6 ] 2+ is 1.0 * 10 9 , what are the...
-
Name each of the following complex ions and identify the oxidation number of the metal: (a) [CrCl 3 (NH 3 ) 2 (OH 2 )] + ; (b) [Rh(en) 3 ] 3+ ; (c) [Fe(Br) 4 (ox)] 3 ; (d) [Ni(OH)(OH 2 ) 5 ] 2+ .
-
When has Equity Theory caused you to lower your efforts in the workplace? Explain how this happened referring to the theory?
-
This year, Druehl, Inc., will produce 60,000 hot water heaters at its plant in Delaware, in order to meet expected global demand. To accomplish this, each laborer at the plant will work 200 hours per...
-
When evaluating qualified plans, it is important to remember that pension plans may best suit employers with unpredictable cash flows due to their discretionary funding feature. When evaluating...

Study smarter with the SolutionInn App


