Calculate the equilibrium constant at 25C and at 100C for each of the following reactions, using data
Question:
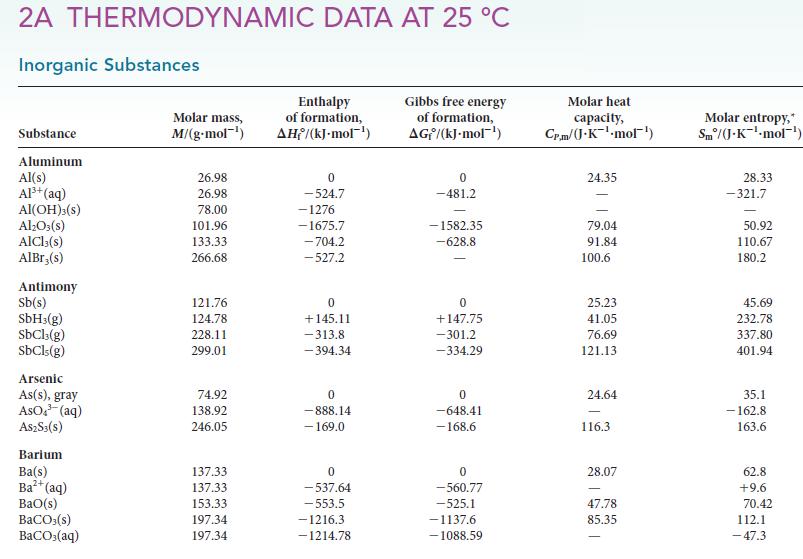
Calculate the equilibrium constant at 25°C and at 100°C for each of the following reactions, using data available in Appendix 2A: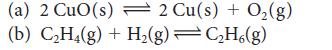
Transcribed Image Text:
(a) 2 CuO (s) 2 Cu(s) + O(g) (b) CH4(g) + H(g) CH6(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Because we want the equilibrium constant at two temperatures we will need to calculate AH and ASo fo...View the full answer

Answered By

Caroline Kinuthia
Taking care of the smaller details in life has a larger impact in our general well being, and that is what i believe in. My name is Carol. Writing is my passion. To me, doing a task is one thing, and delivering results from the task is another thing. I am a perfectionist who always take things seriously and deliver to the best of my knowledge.
4.90+
1933+ Reviews
4269+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
The budgeted income statement for Barnaby's Hideaway is produced on your Excel spreadsheet. Assume that the following constitute the fixed and vari- able costs for the upcoming year Fixed Costs for...
-
Calculate the equilibrium constant Kc at 25oC from the free-energy change for the following reaction: See Appendix C for data. Zn(s) +2Ag (a)Zn2 (a) Ag(s)
-
Calculate the equilibrium constant at 25C and at 150C for each of the following reactions, using data available in Appendix 2A: (a) NH4Cl(s) (b) H(g) + DO(1) NH3(g) + HCl (g) D(g) + HO(1)
-
In Problems 5978, solve each equation in the complex number system. x 2 + 25 = 0
-
Nine 10-? resistors are connected as shown in Figure, and a potential difference of 20 V is applied between points a and b. (a) What is the equivalent resistance of this network? (b) Find the current...
-
The Castillo Products Company described in Problem 6 had a very difficult operating year in 2009, resulting in a net loss of $65,000 on sales of $900,000. In 2010, sales jumped to $1,500,000, and a...
-
Suppose that a $2^{2}$ factorial design studying factors A and B was conducted. An investigator fits the model \[y_{i}=\beta_{0} x_{i 0}+\beta_{1} x_{i 1}+\beta_{2} x_{i 2}+\beta_{3} x_{i...
-
Diamond Software, Inc., does software development. One important activity in software development is writing software code. The manager of the Wordpro Development Team determined that the average...
-
If demand is such that, then P = 40 - 20 a.MR = 400-202 b. MR = 40-40 C. MR 40-Q
-
Three employees of the Horizon Distributing Company will receive annual pension payments from the company when they retire. The employees will receive their annual payments for as long as they live....
-
Determine the temperature change when 4.00 g of (a) KCl; (b) MgBr 2 ; (c) KNO 3 ; (d) NaOH is dissolved in 100. g of water. Assume that the specific heat capacity of the solution is 4.18 J K 1 g 1...
-
When solid NH 4 HS and 0.400 mol NH 3 (g)were placed in a vessel of volume 2.0 L at 24C, the equilibrium NH 4 HS(s) NH 3 (g)+ H 2 S(g), for which Kc = 1.6 * 10 4 , was reached. What are the...
-
In the Boston Museum of Fine Arts is a display of carefully made stone cubes found in the ruins of Mohenjo-Daro of the Indus. The stones are a set of weights that exhibit the binary pattern,...
-
If a stock price follows geometric Brownian motion, what is the process followed by the logarithm of the stock price?
-
What is the purpose of margin accounts?
-
How is the valuation of an option using a binomial tree changed when the stock is assumed to provide a continuous dividend yield?
-
What is the Black-Scholes-Merton price of European call and put options on an asset providing no income?
-
How might investors who consider themselves adept at stock picking use index futures?
-
Your company needs to purchase a track hoe and has narrowed the selection to two pieces of equipment. The first track hoe costs $100,000 and has an hourly operation cost of $31.00 and a useful life...
-
Read the following description and Write a response of it. The discretion of public administrators can be decreased, but not altogether eliminated. Officials will use their discretion in any given...
-
What is the physical origin of the pressure difference across a curved liquidgas interface?
-
Draw the mechanism for each of the following transformations: a. b. c. Br HBr
-
Using the result of Equation (3.8), (P/T) V = / , express as a function of and V m for an ideal gas, and as a function of b, , and V m for a van der Waals gas.
-
1. Define and explain the meaning of a predetermined manufacturing overhead rate that is applied in a job order costing system. ting 2. What are the advantages and disadvantages of using the cost of...
-
Assume that CAPM applies. Suppose that you know that the Stock of a company called " We thank you all for your patience" has a beta equal to 0 . 7 and a risk premium equal to 1 4 % . The risk - free...
-
Can you determine whether the below actions ( i ) to ( v ) can be categorized as a ) Tax Planning, b ) Tax Management, orc ) Tax Evasion, providing reasons for eachi. Mr . Sarthak deposits 1 , 2 5 ,...

Study smarter with the SolutionInn App


