Complete and balance each of the following reactions: (a) XeF (s) + HO(1)XeO3(aq) + HF(aq) (b) Pt(s)
Question:
Complete and balance each of the following reactions: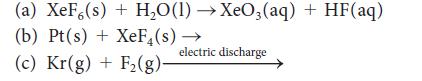
Transcribed Image Text:
(a) XeF (s) + H₂O(1)→XeO3(aq) + HF(aq) (b) Pt(s) + XeF₁(s) (c) Kr(g) + F₂(g)- → electric discharge
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Sure here are the balanced chem...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Complete and balance each of the following molecular equations (in aqueous solution); include phase labels. Then, for each, write the net ionic equation. a. NaOH + HNO3 b. HCl + Ba(OH)2 c. HC2H3O2...
-
Complete and balance each of the following molecular equations (in aqueous solution); include phase labels. Then, for each, write the net ionic equation. a. Al(OH)3 + HCl b. HBr + Sr(OH)2 c....
-
Complete and balance each of the following molecular equations, including phase labels, if a reaction occurs. Then write the net ionic equation. If no reaction occurs, write NR after the arrow. a....
-
Using and citing Bevan, define active empathic listening. Report on both the meaning and significance of the definition Explain how you can use active empathic listening to help you improve your...
-
Here are two useful rules of thumb. The Rule of 72 says that with discrete compounding the time it takes for an investment to double in value is roughly 72/interest rate (in percent). The Rule of 69...
-
Kasey Manufacturing Company started operations on January 1, 2015. During 2015, the company engaged in the following transactions. 1. Issued common stock for $95,000. 2. Paid $33,000 cash to purchase...
-
Complete the second half of the proof of the "forward price formula with carrying cost". To construct the arbitrage, go long one unit of a forward and short one unit spot. To execute the short, it is...
-
Under what circumstances would you consider the use of terms that are high in connotative meaning to be ethical? When would you consider it to be unethical? Explain your reasoning.
-
Consolidated Balance Sheet Working Paper, Bargain Purchase On January 1, 2022, Paxon Corporation acquired all of the outstanding common stock of Saxon Company for $2 billion cash. The prior to the...
-
What justification is there for regarding the ammonium ion as an analog of a Group 1 metal cation? Consider properties such as solubility, charge, and radius. The radius of NH 4 + treated as a...
-
The enthalpy of dissociation of hydrogen bonds, H HBond , is a measure of their strength. Explain the trend seen in the data for the following pure substances, which were measured in the gas phase:
-
An x-ray photon has an energy of 3.3 10 15 J. The frequency that corresponds to this energy is a. 3.3 10 -50 Hz b. 3 10 -19 Hz c. 3.3 10 18 Hz d. 3.3 10 20 Hz
-
A clean wooden box of mass 29[kg] is resting on a clean wooden surface. A person is pushing the box with an increasing force. The force slowly grows from 0, until the force reaches a maximum of 50...
-
Problem 2. National income is 2000, 10% of which is net factor payments from abroad. Taxes are 300, transfers 100, and interest on public debt 100. The government maintains a budget deficit of 5% of...
-
A ring is slipping on a horizontal surface. The center of the ring is moving to the right at 2 m/s. The ring is also rotating counterclockwise at 3 rad/s. What is the angular momentum of the ring...
-
There is growing interest in the extraction of natural gas, whose primary component is methane (CH4), from unusual sources. One such source is natural gas hydrates, where gas molecules are trapped in...
-
The forklift with the dimensions show below have a load of 1700 kg and the forklift itself is 7700 kg. Consider that 4 normal forces are there, 2 at the front wheels (assume they are equal) and 2 at...
-
Targezept Bionics, Inc., has a common stock outstanding that sells for $21 per share. A call option on the stock has an exercise price of $20 and will expire in three months. Based on an analysis of...
-
A fast-food restaurant averages 150 customers per hour. The average processing time per customer is 90 seconds. a. Determine how many cash registers the restaurant should have if it wishes to...
-
In P24.3, the hybrid bonding orbitals for ozone were derived. Use the framework described in Section 24.3 to derive the normalized hybrid lone pair orbital on the central oxygen in O 3 that is...
-
Using your results from Problem P24.10, a. Calculate the s and p character of the water lone pair hybrid orbitals b. Show that the lone pair orbitals are orthogonal to each other and to the hybrid...
-
Use the VSEPR method to predict the structures of the following: a. PF 3 b. CO 2 c. BrF 5 d. SO 2 3
-
You are required to size a shell and tube heat exchanger to cool 100,000 kg/hr of n-heptane at a pressure of 2 bar.g from 95C to 40C before specifying the tube sheet thickness using PD5500. All parts...
-
Which smartphone app is made possible through mobile marketing? Explain.
-
The U . S . Department of Energy's Wind Energy Technologies Office ( WETO ) has looked into many ways to mitigate the impacts and mortality rates of flying animals caused by wind turbines Of the...

Study smarter with the SolutionInn App


