Complete the following table (all values are in kilojoules per mole). Compound, , ion , eg MX
Question:
Complete the following table (all values are in kilojoules per mole).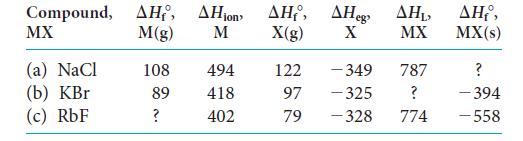
Transcribed Image Text:
Compound, ΔΗ, ΔΗion ΔΗ, ΔΗeg MX M(g) M X(g) X (a) NaCl (b) KBr (c) RbF 108 89 ? 494 418 402 122 97 79 - 349 -325 -328 ΔΗ, MX 787 ? 774 ΔΗ,, MX(s) ? -394 -558
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a 412 kJ ...View the full answer

Answered By

Ankur Gupta
I have a degree in finance from a well-renowned university and I have been working in the financial industry for over 10 years now. I have a lot of experience in financial management, and I have been teaching financial management courses at the university level for the past 5 years. I am extremely passionate about helping students learn and understand financial management, and I firmly believe that I have the necessary skills and knowledge to effectively tutor students in this subject.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Compute the amount of the gift in the following independent scenarios. If an amount is zero, enter "0". a. Bolivar and Collen purchase real estate as tenants in common. Of the $250,000 purchase...
-
Which of the following would produce a materials price variance? abreakage of materials in production. ban excess quantity of materials used can excess number of direct labor-hours worked in...
-
Calculate a lattice energy for CaH2(s) in kilojoules per mole using the following information: (a) Eea for H(g) = -72.8kj/mol. (b) Ei1 for Ca(g) = +589.8kJ/mol. (c) Ei2 for Ca(g) = +1154kJ/mol. Heat...
-
Chemistry A one-electron atom is an atom with Z protons in the nucleus and one electron. For example, Z = 2 for helium and Z = 3 for lithium. Use our class discussion of the allowed radii and...
-
G-Bar Pastures is a 400-acre farm on the outskirts of the Kentucky Bluegrass, specializing in the boarding of broodmares and their foals. A recent economic downturn in the thoroughbred industry has...
-
Sketch the curves of the given trigonometric functions. Check each using a calculator. y = 5 cos (4)
-
Imagine the following scenario: For some time, you have known that you need to shop for a new mattress. You are putting off doing this because you dread the experience. You imagine yourself walking...
-
1. In what ways does UTCs corporate- level strategy of unrelated diversification create value? 2. What are the dangers and disadvantages of this strategy? 3. Collect some recent information on UTC...
-
1 What is HRM 2 What is Training and Developmnt 3 What is Recruitment 4 What is Selection 5 What is HRP 6 What is Job Analysis 7 What is a Career Planning and Development 8 What is Job-Description 9...
-
Air in a bicycle pump is compressed by pushing in the handle. The inner diameter of the pump is 3.0 cm and the pump is depressed 20. cm with a pressure of 2.00 atm. (a) How much work is done in the...
-
Which substance in each of the following pairs has the higher molar entropy? (Take the temperature to be 298 K unless otherwise specified.) (a) O 2 (g) or O 3 (g); (b) CH 2 Br 2 (g) or CH 4 (g); (c)...
-
A space station sounds an alert signal at time intervals of 1. 00 h. Spaceships A and B pass the station, both moving at \(0.400 c_{0}\) relative to the station but in opposite directions. How long...
-
Provide an example of a good externality, that is, one that increases a projects true NPV.
-
Jay is a limited partner in Kappa Sales, a limited partnership. Jay is liable for the firms debts a. in no way. b. in proportion to the total number of partners in the firm. c. to the extent of his...
-
What is one classification scheme that firms often use to obtain risk-adjusted costs of capital?
-
How can stock repurchases help a company operate in accordance with the residual dividend model?
-
Define the following terms: a. Capital budgeting; strategic business plan b. Net present value (NPV) c. Internal rate of return (IRR) d. NPV profile; crossover rate e. Mutually exclusive projects;...
-
Which of the following goods are scarce? a. Garbage b. Salt water in the ocean c. Clothes d. Clean air in a big city e. Dirty air in a big city f. A public library
-
SBS Company have received a contract to supply its product to a Health Care Service Hospital. The sales involve supplying 1,250 units every quarter, the sales price is RM 85 per unit. The Client...
-
The Ostwald process for the commercial production of nitric acid involves three steps: 2NO(g) + O2(g) 2NO2(g) 3NO2(g) + H2O(l) 2HNO3(l) + NO(g) a. Calculate ÎHo, ÎSo, ÎGo, and K (at...
-
Consider the following reaction at 800. K: N2(g) + 3F2(g) 2NF3(g) An equilibrium mixture contains the following partial pressures: PN2 = 0.021 atm, PF2 = 0.063 atm, and PNF3 = 0.48 atm. Calculate Go...
-
Consider the following reaction at 298 K: 2SO2(g) + O2(g) 2SO3(g) An equilibrium mixture contains O2(g) and SO3(g) at partial pressures of 0.50 atm and 2.0 atm, respectively. Using data from...
-
Shown below is the stockholders' equity section of Holiday Travels' balance sheet at December 31, 2023: Common stock, $3 par value, 5,000,000 shares authorized, 330,000 shares issued and outstanding...
-
A company set its predetermined overhead rate at the beginning of the year using the following estimates: overhead costs, $2,243,780, and direct labor costs, $434,000. Actual costs for the year are...
-
Evander bought a house on June 1, 2021. The previous owners had been foreclosed on, and Evander was able to buy the house below market, for $155,000. He made no improvements while he owned it. On...

Study smarter with the SolutionInn App


