Consider the following data: Co+ + e- Co(en) 3+ Co(en)33+ Co+ 8 = 1.82 V Kf =
Question:
Consider the following data:
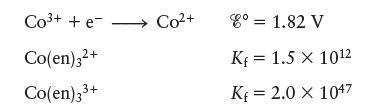
Transcribed Image Text:
Co³+ + e- Co(en) 3²+ Co(en)33+ Co²+ 8⁰ = 1.82 V Kf = 1.5 X 1012 Kf = 2.0 X 1047
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
ANSWER It appears that the given data represents two different equilibrium reactions involving cobal...View the full answer

Answered By

Brian Kiprono
Taking part in public speeches, blogging, writing essays
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following data for two products of Rowena Manufacturing. Required 1. Using direct labor hours as the basis for assigning overhead costs, determine the total production cost per unit for...
-
Consider the following data from two independent samples. These data can also be found in the Excel file Prob1813.xlsx. Perform a hypothesis test using α = 0.05 to determine if the...
-
Consider the following data from two independent samples. These data can also be found in the Excel file Prob1814.xlsx. Perform a hypothesis test using α = 0.10 to determine if the...
-
Suppose you want to buy a house that is sold by way of a first-price sealed bid auction. In contrast to the model in the lecture, there are more than 2 players. Players simultaneously and...
-
Using random digit dialing, researchers call 1400 people and ask what obstacles (such as childcare) keep them from exercising. Identify the sampling technique used, and discuss potential sources of...
-
Crate A is traveling down the incline with a speed of 4 m/s when in the position shown. It later strikes and becomes attached to crate B. Determine the distance d moved by the pair after the...
-
FRAUD PREVENTION AND DETERRENCE IN ACTION Assume the following facts: thirty-day collection period. The invoice is used to post-sales to the accounting system. Checks are received in the mailroom and...
-
Sawaya Co., Ltd., of Japan is a manufacturing company whose total factory overhead costs fluctuate considerably from year to year according to increases and decreases in the number of direct...
-
Question 17 (1 point) If the credit to record the payment of an account payable is not posted Liabilities will be understated Expenses will be understated Cash will be overstated Which statement is...
-
Design the capacity of a fuel tank for a petrol fuelled Passenger Auto-Rickshaw. For details of the vehicle, you can use the system specifications of a commercially available auto rickshaw in the...
-
What is the hybridization of the underlined nitrogen atom in each of the following molecules or ions? a. NO - b. N 2 O 3 (O 2 NNO) c. NO 2 - d. N 2
-
Given the following data: P4(s) + 6Cl(g) 4PC13(g) P4(s) + 50(g) P4010(S) PC13(g) + Cl(g) PCls(g) PC3(g) + O(g) ClPO(g) calculate AH for the reaction AH-1225.6 kJ AH = -2967.3 kJ = 84.2 kJ AH =...
-
Our treatment of the electroweak interactions of massless leptons may be extended to massless quarks by invoking leptonquark symmetry between the doublets (ν e , e ) and (u, d) where we...
-
How does ethnic identity intersect with broader social constructs such as race and nationality in contemporary societies?
-
What are some of the benefits of juvenile courts? Are there any arguments to be made against them?
-
What is "invisible blackness"? Why was it of such concern to those who supported racial classification laws?
-
How did the U.S. Constitution and laws affect the development of the U.S. courts?
-
How does the concept of "ethnicity" differ across various academic disciplines such as anthropology, sociology, and political science, and what implications does this interdisciplinary variation have...
-
The speed in which the home page of a website is downloaded is an important quality characteristic of that website. Suppose that the mean time to download the home page for the Internal Revenue...
-
Explain how two samples can have the same mean but different standard deviations. Draw a bar graph that shows the two samples, their means an standard deviations as error bars. T S
-
Use the accompanying phase diagram for carbon to answer the following questions. a. How many triple points are in the phase diagram? b. What phases can coexist at each triple point? c. What happens...
-
Iodine, like most substances, exhibits only three phases: solid, liquid, and vapor. The triple point of iodine is at 90 torr and 115oC. Which of the following statements concerning liquid I2 must be...
-
The melting point of a fictional substance X is 225oC at 10.0 atm. If the density of the solid phase of X is 2.67 g/cm3 and the density of the liquid phase is 2.78 g/cm3 at 10.0 atm, predict whether...
-
Draw quadrilateral A(2,1)B(4,2)C(5,5)D(1,3). Use a scale factor of 2 to draw a dilation of the quadrilateral.
-
A 100-degree arc of a circle has a length of 7cm. To the nearest centimeter, what is the radius of the circle?
-
Calculate activity rates for each activity cost pool using activity-based costing (ABC). (10%) Determine the amount of overhead cost that would be assigned to each product using activity-based...

Study smarter with the SolutionInn App


