Consider the following reaction to produce methyl acetate: When this reaction is carried out with CH3OH containing
Question:
Consider the following reaction to produce methyl acetate: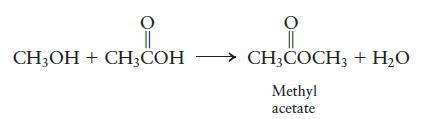
When this reaction is carried out with CH3OH containing radioactive oxygen-18, the water produced is not radioactive. Explain.
Transcribed Image Text:
CH3OH + CH3COH CH3COCH3 + H₂O Methyl acetate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
My explanation is that the water that is formed during the react...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following reaction to produce methyl acetate: When this reaction is carried out with CH3OH containing radioactive oxygen-18, the water produced is not radioactive. Explain. CH,OH CH...
-
Consider the reaction to produce the ester methyl acetate: When this reaction is carried our with CH3OH contain¬ing radioactive oxygen-18, the water produced does not contain oxygen-18. Explain...
-
When (S)-2-bromopropanoic acid [(S)-CH3CHBrCO2H] reacts with concentrated sodium hydroxide, the product formed (after acidification) is (R)-2-hydroxypropanoic acid [(R)-CH3CHOHCO2H, commonly known as...
-
A partially completed flowchart showing some of the major documents commonly used in the purchasing function of a merchandise business is presented below. Identify documents 1, 3, and4. Purchase Order
-
Use the frequency distribution from Try It Yourself 2 to construct a frequency polygon that represents the ages of the 50 most powerful women listed on page 39. Describe any patterns. 26, 31, 35, 37,...
-
The system is released from rest with no slack in the cable and with the spring unstretched. Determine the distance s traveled by the 4-kg cart before it comes to rest (a) If m approaches zero (b) If...
-
Reconsider the data from Problem 4. Management has expressed some concern over the life of the project and the impact of possible early termination. As a result, you have developed additional data...
-
Assume that you are preparing for a second interview with a manufacturing company. The company is impressed with your credentials but has indicated that it has several qualified applicants. You...
-
Sketch the following set (shade the region inside the set): {x R | ||Ax b|| 1}, where b = H and A = [ ]
-
Cincinnati Flow Technology (CFT) has purchased 10,000 pumps annually from Kobec. Inc. Because the price keeps increasing and reached $ 102.00 per unit last year, CF'T's management has asked for an...
-
Iodine-131 has a half-life of 8.0 days. How many days will it take for 174 g of 131 I to decay to 83 g of 131 I?
-
How could a radioactive nuclide be used to demonstrate that chemical equilibrium is a dynamic process?
-
Evaluate where S is the hemisphere x 2 + y 2 + z 2 = 1, z 0. [[ ' S VX (yi). n do,
-
If the real rate of interest is 1.75% and if the expected inflation rate is 8.65%, what is the present value of $50,000 to be received exactly 8 years from today? 2. You expect to deposit the...
-
What is the value today of a money machine that will pay $1,340.00 every six months for 16.00 years? Assume the first payment is made 1.00 years from today and the interest rate is 14.00%. What is...
-
A topic that has a local focus, such as a change that needs to happen in your community. Please include three things: what the need for change is, what your planned solution is, and who the audience...
-
Derek will deposit $849.00 per year for 12.00 years into an account that earns 8.00%. The first deposit is made today. How much will be in the account 12.0 years from today? Note that he makes 12.0...
-
There is a need for a parking lot within a given town. A land in a suitable location is available for purchase that they could pave to provide some additional parking. Based on a survey of local...
-
Given a normal distribution with = 100 and = 10, if you select a sample of n = 25, what is the probability that is a. Less than 95? b. Between 95 and 97.5? c. Above 102.2? d. There is a 65% chance...
-
Difference between truncate & delete
-
Describe, in general, the structures of ionic solids. Com-pare and contrast the structures of sodium chloride and zinc sulfide. How many tetrahedral holes and octahedral holes are there per closest...
-
Assume the two-dimensional structure of an ionic com-pound MxAy is What is the empirical formula of this ionic compound?
-
Identify the most important types of interparticle forces present in the solids of each of the following substances. a. Ar b. HCl c. HF d. CaCl2 e. CH4 f. CO g. NaNO3 h. NH4Cl i. Teflon,...
-
There is a polynomial that has three roots. One root is 7.5 , one root is 1+2i. What is the third root?
-
To practice strategizing how to solve systems of equations using the elimination method, consider these two equations: x+2y-z=2 x-y+3z=8.
-
Given the volume of a cuboid can be represented by the expression 5x(15-2x)^(2). Calculate the volume of the cuboid.

Study smarter with the SolutionInn App


