Decide on the basis of the information in Table 6C.3 whether carbonic acid is a strong or
Question:
Decide on the basis of the information in Table 6C.3 whether carbonic acid is a strong or weak acid in liquid ammonia solvent. Explain your answer.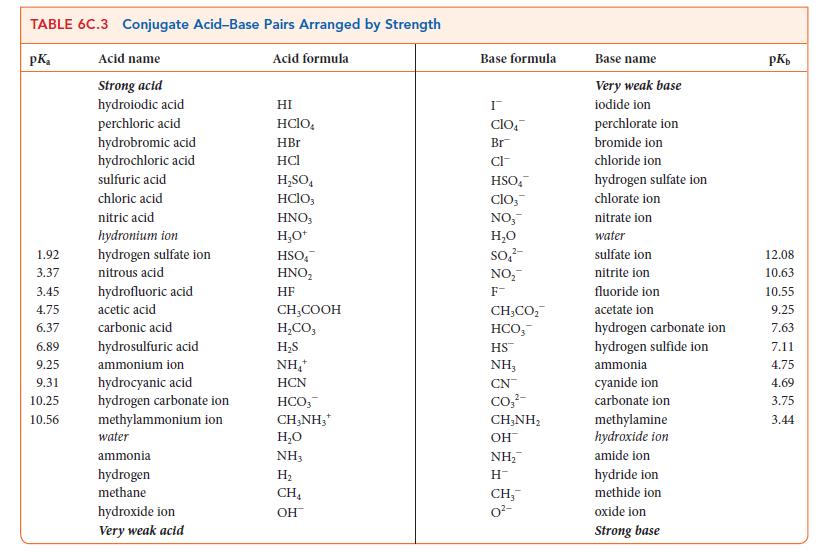
Transcribed Image Text:
TABLE 6C.3 Conjugate Acid-Base Pairs Arranged by Strength pK₂ 1.92 3.37 3.45 4.75 6.37 6.89 9.25 9.31 10.25 10.56 Acid name Strong acid hydroiodic acid perchloric acid hydrobromic acid hydrochloric acid sulfuric acid chloric acid nitric acid hydronium ion hydrogen sulfate ion nitrous acid hydrofluoric acid acetic acid carbonic acid hydrosulfuric acid ammonium ion hydrocyanic acid hydrogen carbonate ion methylammonium ion water ammonia hydrogen methane hydroxide ion Very weak acid Acid formula HI HCIO. HBr HCI H₂SO4 HClO3 HNO3 H₂O+ HSOA HNO₂ HF CH₂COOH H,CO, H₂S NH₂+ HCN HCO, CH3NH₂* H₂O NH3 H₂ CH₁ OH Base formula I clo Br CI- HSOA Clo, NO, H₂O SO₂² NO₂™ F CH,CO, HCO, HS™ NH, CN CO₂²- CHÍNH, OH NH₂ H™ CH₂™ 0²- Base name Very weak base iodide ion perchlorate ion bromide ion chloride ion hydrogen sulfate ion chlorate ion nitrate ion water sulfate ion nitrite ion fluoride ion acetate ion hydrogen carbonate ion hydrogen sulfide ion ammonia cyanide ion carbonate ion methylamine hydroxide ion amide ion hydride ion methide ion oxide ion Strong base pKb 12.08 10.63 10.55 9.25 7.63 7.11 4.75 4.69 3.75 3.44
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Based on the information in Table 6C3 carbonic acid is a weak acid in liquid ammonia solvent Heres w...View the full answer

Answered By

Hemstone Ouma
"Hi there! My name is Hemstone Ouma and I am a computer scientist with a strong background in hands-on experience skills such as programming, sofware development and testing to name just a few. I have a degree in computer science from Dedan Kimathi University of Technology and a Masters degree from the University of Nairobi in Business Education. I have spent the past 6 years working in the field, gaining a wide range of skills and knowledge. In my current role as a programmer, I have had the opportunity to work on a variety of projects and have developed a strong understanding of several programming languages such as python, java, C++, C# and Javascript.
In addition to my professional experience, I also have a passion for teaching and helping others to learn. I have experience as a tutor, both in a formal setting and on a one-on-one basis, and have a proven track record of helping students to succeed. I believe that with the right guidance and support, anyone can learn and excel in computer science.
I am excited to bring my skills and experience to a new opportunity and am always looking for ways to make an impact and grow as a professional. I am confident that my hands-on experience as a computer scientist and tutor make me a strong candidate for any role and I am excited to see where my career will take me next.
5.00+
8+ Reviews
22+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Acetic acid is used as a solvent for some reactions between acids and bases. (a) Nitrous acid and carbonic acids are both weak acids in water. Will either of them act as a strong acid in acetic acid?...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
A statistician has to decide on the basis of one observation whether the parameter of a Bernoulli distribution is 0, 1/2 , or 1; her loss in dollars (a penalty that is deducted from her fee) is 100...
-
The following rates are observed on the yield curve: 6-month 0.5%, 12-month 1.5%, 18-month 2%, 24-month 3%. You hold a portfolio with four semi-annual coupon bonds, all with a par-value of $1,000 and...
-
Support-department cost allocation, reciprocal method (continuation of 15-19). Refer to the data given in Exercise 15-19. 1. Allocate the two support departments costs to the two operating...
-
With the growth of Hard Rock Caffrom one pub in London in 1971 to more than 129 restaurants in more than 40 countries todaycame a corporate wide demand for better forecasting. Hard Rock uses...
-
What are the final two steps a researcher should do after presenting the research findings to the decision makers?
-
Faber Company had gross wages of $110,000 during the week ended June 17. The amount of wages subject to social security tax was $110,000, while the amount of wages subject to federal and state...
-
What is the process used when replacing a fund manager. What are the issues that need to be considered in the process? Discuss the considerations and problems in replacing a manager and the costs...
-
(a) Complete the following table. (b) Rank the four solutions in order of increasing acidity. [H3O+] (i) 0.50 mol-L- (ii) (iii) (iv) [OH-] -1 0.50 mol-L- pH -0.10 POH -0.10
-
You wish to separate barium ions from calcium ions by selective precipitation. Which anion, fluoride or carbonate, would be the better choice for achieving this precipitation? Why?
-
From a molecular perspective, briefly explain the mechanism by which clay minerals become hydroplastic when water is added.
-
In 1957, Procter & Gamble acquired Green Bay, Wisconsin-based tissue maker Charmin, its first consumer-paper products business. One product in particular, Charmin Towels, was a single-ply towel that...
-
Blossom Inc's net sales and gross profit were $333400 and $120150 respectively. Assuming the cost of goods available for sale were $269600, what was the cost value of the ending imentory?
-
Using the data set CarPrice, carry out the following question: a. Construct a table that show the sample mean, standard deviation, and minimum and maximum values for each variable. b. Construct a...
-
On January 1 , 2 0 X 3 ANL Pte Ltd paid $ 2 6 , 0 0 0 , 0 0 0 to purchase a building for its office use. The useful life of the building is 5 0 years with zero residual value. The company financial...
-
Suppose that S = {u, 12, 13, 14} where 72 56 u = 23 199 -144 -113 197 u = -299 -187 60 244 -290 u3 = 72, 23, 199, -144, -113, 197 56, -299, -187, 60, 244, -290 291, 267, -233, -13, -95, 179 -201, 18,...
-
Yum Yum makes jams and other preserves from seasonal fruits and berries. The production process begins with the berries and sugar. After processing, Yum Yum packs the finished jams into glass...
-
The percentage of completion and completed contract methods are described in the FASB ASC. Search the codification to find the paragraphs covering these topics, cite them, and copy the results.
-
The dissolution of 7.75g of a substance in 825 g of benzene at 298 K raises the boiling point by 0.575C. Note that K f = 5.12 K kg mol 1 , K b = 2.53 K kg mol 1 , and the density of benzene is 876.6...
-
Describe what you would observe if you heated the solid at the composition 40. atomic percent Si in Figure 9.26 from 300.C to 1300.C.
-
An ideal dilute solution is formed by dissolving the solute A in the solvent B. Write expressions equivalent to Equations (9.9) through (9.13) for this case.
-
Consider an individual who immigrates to Canada and deposits $5,000 into the Canadian banking system. Suppose that all commercial banks have a target reserve ratio of 5 percent and that individuals...
-
1. Name and explain at least three unspoken "rules" or norms regarding nonverbal communication in your culture, family, friend group, etc. 2. Share a picture or short video clip (keep it rated PG) of...
-
You are head of the marketing department at BTS Ltd, a major electrical appliance manufacturer in Australia. Your recent social media monitoring efforts pick up substantial activity where customers...

Study smarter with the SolutionInn App


