Determine the formal charge on each atom in the following molecules. Identify the structure of lower energy
Question:
Determine the formal charge on each atom in the following molecules. Identify the structure of lower energy in each pair.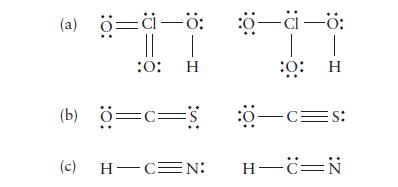
Transcribed Image Text:
(a) ö=ä-ö: || | :0: H (b) Ö=c=S (c) H-C=N: :0—a—ö: T :0: :8-c=s: | H H-CIN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
0 0 a CI b c O H 0 lo...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Determine the formal charge on each atom in the following ions. Identify the structure of lowest energy in each case. (a) N=c= -: (9) (c) :: 0: :0: 7- :: :N=C- :O: As :0: :: 7- = :0: :O: =As :0: ::...
-
Two contributions to the resonance structure are shown below for each species. Determine the formal charge on each atom and then, if possible, identify the Lewis structure of lower energy for each...
-
Determine the formal charge on each atom in the following molecules and ions: (a) SO 2 (b) OSCl 2 (c) O 2 SCl 2 (d) FSO 3
-
Which of the following represents the GREATEST risk created by a reciprocal agreement for disaster recovery made between two companies? A. Developments may result in hardware and software...
-
The investment committee of Grid Iron Restaurants Inc. is evaluating two restaurant sites. The sites have different useful lives, but each requires an investment of $565,000. The estimated net cash...
-
What is a shill bidder in an online auction?
-
You are the technical accounting consultant of a professional accounting firm. One of your clients is an Australian travel company that arranges package tours to overseas destinations. The client...
-
Selected information from the payroll register of Howard's Cutlery for the week ended October 7, 20--, is presented below. Social Security tax is 6.2% on the first $106,800 of earnings for each...
-
(a) NP and Co. has imported goods for US $ 7,00,000. The amount is payable after three months. The company has also exported goods for US $ 4,50,000 and this amount is receivable in two months. For...
-
Structural isomers are molecules that have the same composition but a different pattern of connectivity. Two isomers of disulfur difluoride, S 2 F 2 , are known. In each, the two S atoms are bonded...
-
Give the ground-state electron configuration and number of unpaired electrons expected for each of the following ions: (a) Fe 3+ ; (b) Bi 3+ ; (c) Si 4+ ; (d) I 2 .
-
In problem, information is given about a polynomial function f(x) whose coefficients are real numbers. Find the remaining zeros of f. Degree 3; zeros: 3, 4 - i
-
If requirements are frequently changing, which of the following models is to be selected? (a) Waterfall model (b) Prototyping model (c) RAD model (d) Agile mode
-
If requirements are easily understandable and defined, which of the following models is best suited? (a) Waterfall model (b) Prototyping model (c) Spiral model (d) None of these
-
Which of the following are new introductions in UML 2? (a) Guards (b) Combined fragments (c) Interaction Occurrences (d) Frames
-
Waterfall model is not suitable for (a) Small projects (b) Accommodating change (c) Complex projects (d) None of these
-
Actor Bank has an association with use case Withdraw with multiplicity 0..*. This means that Bank is involved with multiple Withdraw operations (a) sequentially (b) parallely (c) either (d) neither
-
True or False: If a monopolist owned an exhaustible resource, he would control its availability so that the price rose faster than the rate of interest.
-
Gordon and Lisa estimate that they will need $1,875,000 in 40 years for their retirement years. If they can earn 8 percent annually on their funds, how much do they need to save annually?
-
The deletion of a single base from a DNA molecule can be a fatal mutation. Substitution of one base for another is often not as serious a mutation. Why?
-
Draw the structures of the tripeptides gly-ala-ser and serala-gly. How many other tripeptides are possible using these three amino acids?
-
Aspartame, the artificial sweetener marketed under the name NutraSweet, is a methyl ester of a dipeptide. The structure of aspartame is a. What two amino acids are used to prepare aspartame? b. There...
-
Consider a lake, in which the residence time of water is 10 years. Into this lake a highly soluble, non-co-distilling pollutant enters at a constant rate, a, and out of the lake pollutant exits, in...
-
Tasteez is preparing their 2018 budget. They want to prepare a flexible budget for use each month. They estimate sales/production will be between 1,500,000 and 2,000,000 boxes of frozen TV dinners...
-
Note: creat a survey using qulatrics Based on the research objectives provided below, you need to... Based on the research objectives I have provided below, you need to think about possible questions...

Study smarter with the SolutionInn App


