Determine which of the following compounds are stable with respect to decomposition into their elements under standard
Question:
Determine which of the following compounds are stable with respect to decomposition into their elements under standard conditions at 25°C (see Appendix 2A):
(a) C3H6(g), cyclopropane;
(b) CaO(s);
(c) N2O(g);
(d) HN3(g).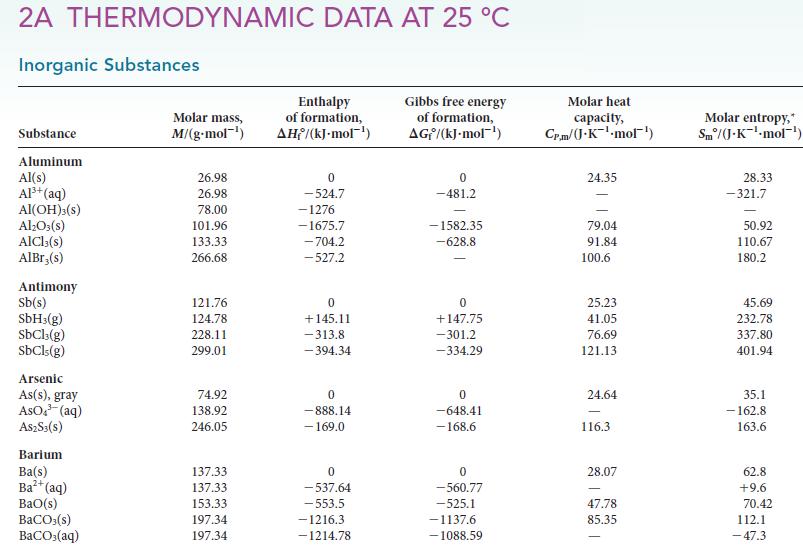
Transcribed Image Text:
2A THERMODYNAMIC DATA AT 25 °C Inorganic Substances Substance Aluminum Al(s) Al³+ (aq) Al(OH)3(S) Al₂O3(s) AlCl3(s) AlBr,(s) Antimony Sb(s) SbH3(g) SbCl3(g) SbCls (g) Arsenic As(s), gray AsO³(aq) A$2S3(S) Barium Ba(s) Ba²+ (aq) BaO(s) BaCO3(s) BaCO3(aq) Molar mass, M/(g.mol-¹) 26.98 26.98 78.00 101.96 133.33 266.68 121.76 124.78 228.11 299.01 74.92 138.92 246.05 137.33 137.33 153.33 197.34 197.34 Enthalpy of formation, AH/(kJ-mol-¹) 0 -524.7 -1276 -1675.7 -704.2 -527.2 0 +145.11 -313.8 -394.34 0 -888.14 - 169.0 0 -537.64 -553.5 -1216.3 -1214.78 Gibbs free energy of formation, AG/(kJ.mol-¹) 0 -481.2 -1582.35 -628.8 0 +147.75 -301.2 -334.29 0 -648.41 -168.6 0 -560.77 -525.1 -1137.6 -1088.59 Molar heat capacity, Cr.m/J.K¹-mol¹) 24.35 79.04 91.84 100.6 25.23 41.05 76.69 121.13 24.64 116.3 28.07 47.78 85.35 Molar entropy,* Sm/(J-K¹-mol-¹) 28.33 -321.7 50.92 110.67 180.2 45.69 232.78 337.80 401.94 35.1 -162.8 163.6 62.8 +9.6 70.42 112.1 -47.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To evaluate whether the compounds are stable with respect to decomposition into their elements under ...View the full answer

Answered By

Hemstone Ouma
"Hi there! My name is Hemstone Ouma and I am a computer scientist with a strong background in hands-on experience skills such as programming, sofware development and testing to name just a few. I have a degree in computer science from Dedan Kimathi University of Technology and a Masters degree from the University of Nairobi in Business Education. I have spent the past 6 years working in the field, gaining a wide range of skills and knowledge. In my current role as a programmer, I have had the opportunity to work on a variety of projects and have developed a strong understanding of several programming languages such as python, java, C++, C# and Javascript.
In addition to my professional experience, I also have a passion for teaching and helping others to learn. I have experience as a tutor, both in a formal setting and on a one-on-one basis, and have a proven track record of helping students to succeed. I believe that with the right guidance and support, anyone can learn and excel in computer science.
I am excited to bring my skills and experience to a new opportunity and am always looking for ways to make an impact and grow as a professional. I am confident that my hands-on experience as a computer scientist and tutor make me a strong candidate for any role and I am excited to see where my career will take me next.
5.00+
8+ Reviews
22+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Determine which of the following compounds are stable with respect to decomposition into their elements under standard conditions at 25C (see Appendix 2A): (a) PCl 5 (g); (b) HCN(g); (c) NO(g); (d)...
-
The term thermodynamic stability refers to the sign of r G . If r G is negative, the compound is stable with respect to decomposition into its elements. Use the data in Appendix D to determine...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Using the following uniform density curve, answer the question. Plx) 125 7 8 What is the probability that the random variable has a value less than 5? 0.125 0.625 K0.75 0.5
-
A straight-line charge with constant linear charge density ? is located perpendicular to the x-y plane in the first quadrant at (x 0 , y 0 ). The intersecting planes x = 0, ? ? 0 and ? = 0, x ? 0 are...
-
What is IVR? How is it used to query a database?
-
A bullet of speed \(v\) and inertia \(m\) strikes and gets embedded in a wooden block of inertia \(M\), initially at rest on a low-friction floor. Plot \(k_{f} / k_{i}\) as a function of \(M / m\)...
-
Amortization schedule for bonds. On January 1, 2008, Seward Corporation issues $100,000 face value, 8% semiannual coupon bonds maturing three years from the date of issue. The coupons, dated for June...
-
7. The full symbol of an ion of an element is 39X+? Which one of the following is the number of electrons in the ion? A. 18 B. 19 C. 20 D. 39 8. The atomic numbers of elements W, X, Y and Z are 9,...
-
Robert Curl, Richard Smalley, and Harold Kroto were awarded the Nobel Prize in Chemistry in 1996 for the discovery of the soccer ball shaped molecule C 60 . This molecule was the first of a new...
-
Draw the Lewis structure for the hypothetical molecule N 6 , consisting of a six-membered ring of nitrogen atoms. Using bond enthalpies, calculate the enthalpy of reaction for the decomposition of N...
-
Write each fraction as a percent. 7 50
-
Describe three audit procedures related to each of the three triple constraints model elements.
-
Explain the triple constraints model and its implications for effective project management.
-
List five continuous monitoring/auditing routines you would recommend to a client for a customer call center facility. Specify what data would be collected, how it would be analyzed, the reporting...
-
List three reasons project planning should include a risk assessment.
-
What are the five phases of projects? List three operational audit steps for each of them.
-
In 1976, the parents of a seven-year-old boy sued a New York hospital for $3.5 million. The boy was blinded shortly after he was born two weeks premature. His parents claimed that hospital doctors...
-
As you rewrite these sentences, replace the cliches and buzzwords with plain language (if you don't recognize any of these terms, you can find definitions online): a. Being a jack-of-all-trades, Dave...
-
Super glue contains methyl cyanoacrylate which readily polymerizes on exposure to traces of water or alcohols on the surfaces to be bonded together. The polymer provides a strong bond between the two...
-
Super glue contains methyl cyanoacrylate which readily polymerizes on exposure to traces of water or alcohols on the surfaces to be bonded together. The polymer provides a strong bond between the two...
-
The polyester formed from lactic acid, is used for tissue implants and surgical sutures that will dissolve in the body. Draw the structure of a portion of this polymer.
-
Problem 2 For the beam and cross section shown, draw the SFD and BMD; identify the largest positive and negative bending moments; then, compute the maximum load q that can be applied to the beam...
-
If the gymnast standing at rest creates a force = 4 5 0 N and the total impulse of the jump is 9 8 0 Ns over a time of 2 . 1 second, find the following: impulse due to weight during the whole...
-
A motor draws 42 A at full load and is operated at continuous duty. All motor starter terminations are rated at 75 C. What is the minimum branch circuit RW90 copper conductor size? AWG/kcmil What is...

Study smarter with the SolutionInn App


