Does the information on alkali metals in Table 12.9 of the text confirm the general periodic trends
Question:
Does the information on alkali metals in Table 12.9 of the text confirm the general periodic trends in ionization energy and atomic radius? Explain.
Table 12.9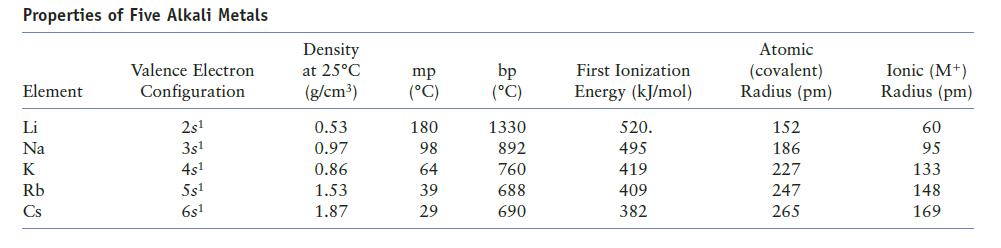
Transcribed Image Text:
Properties of Five Alkali Metals Element Li Na K Rb Cs Valence Electron Configuration 2s¹ 3s¹ 4s¹ 5s¹ 6s1 Density at 25°C (g/cm³) 0.53 0.97 0.86 1.53 1.87 mp (°C) 180 98 64 39 29 bp (°C) 1330 892 760 688 690 First Ionization Energy (kJ/mol) 520. 495 419 409 382 Atomic (covalent) Radius (pm) 152 186 227 247 265 Ionic (M+) Radius (pm) 60 95 133 148 169
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Answer The information on alkali metals in Table 129 does confirm the general ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The radius trend and the ionization energy trend are exact opposites. Does this make sense? Define electron affinity. Electron affinity values are both exothermic (negative) and endothermic...
-
Explain why the first ionization energy tends to increase as one proceeds from left to right across a period. Why is the first ionization energy of aluminum lower than that of magnesium and the first...
-
The ionization energy of O2 is smaller than the ionization energy of atomic O; the opposite is true for the ionization energies of N2 and atomic N. Explain this behavior in terms of the molecular...
-
a. Consider the general effect of the discount rate on the dynamic efficient allocation of a depletable resource across time. Suppose we have two versions of the two-period model discussed in this...
-
A veterinarian records the weights of dogs treated at a clinic. The weights are normally distributed, with a mean of 52 pounds and a standard deviation of 15 pounds. Find the weights x corresponding...
-
Assuming that list is a List, explain in detail the stream pipeline: list.stream() .filter(value -> value % 2 != 0) .reduce(0, Integer::sum)
-
Its not like what youve done has really hurt anyone. At most, the missing money might cost earnings-pershare, maybe a penny. Is that what caused you to write those checks in the name of your company?
-
1. Prepare a context diagram for the new system. 2. Prepare a diagram 0 DFD for the new system. 3. Write a brief memo that explains at least five attributes that you can use to define a process in...
-
Bleeker Street Pizza and Keste Pizza & Vino are located close to each other on Bleeker Street in the West Village. There are 450 potential customers every day, and suppose that each of them is...
-
The purpose of this assignment is to analyze how businesses interact with their internal and external environments. Students will also apply their knowledge of decision-making models, technology and...
-
Spectroscopists use emission spectra to confirm the presence of an element in materials of unknown composition. How is this possible?
-
The electron affinity for sulfur is more exothermic than that for oxygen. How do your account for this?
-
The United States uses approximately 3.0 trillion kWh of electricity annually. If 20% of this electrical energy were supplied by nuclear generating plants, how much nuclear mass would have to be...
-
The capital accounts of Trent Henry and Tim Chou have balances of $ 1 3 6 , 5 0 0 and $ 9 5 , 5 0 0 , respectively. LeAnne Gilbert and Becky Clarke are to be admitted to the partnership. Gilbert buys...
-
When the Montreal Gazette newspaper reduced the weekly subscription price from $5.00 to $4.00 (a savings of 20), the number of subscriptions sold increased by 5%. This suggests that their customers...
-
Pottery Barns in house team of economists has estimated the following U.S. market demand function for Pottery Barns iconic apothecary table (which is given below). Let good X be the apothecary table...
-
BUDGET For example, the discrepancy in local promotion expenses may have been a legitimate result of a one-off approval taken from Head Office and have caused an unfavourable variance of $50,000. The...
-
Suppose a country's nominal GDP is $600 billion, government expenditures less debt service are $155 billion, and revenues are $170 billion. The nominal debt is $360 billion. Inflation is 5 percent...
-
Refer to the results in Problem 16.15 on page 682 and Problem 16.27 on page 692 concerning the number of barrels of oil in the U.S. strategic oil reserve (stored in Strategic). a. Perform a residual...
-
You are standing on the top of a building and throw a ball vertically upward. After 2 seconds, the ball passes you on the way down, and 2 seconds after that, it hits the ground below. a. What is the...
-
You are told that 2.55 g of a gaseous hydrocarbon occupies a vessel of volume 3.00 L at 0.950 atm and 82.0C. Draw the Lewis structure of this hydrocarbon.
-
A chemist prepares a sample of hydrogen bromide and finds that it occupies 500. mL at 45C and 120. Torr. What volume would it occupy at 0 C at the same pressure?
-
The expression for the capillary rise in Exercise 3G.11 assumes that the tube is vertical. How will the expression be modified when the tube is held at an angle (theta) to the vertical? Exercise...
-
Oliver Clone is a 23 year old man who is not married. Oliver has one nephew, Sammy Jones who he is very close to. Oliver had a good weekend in Vegas and won $300,000 at poker. Oliver deposits the...
-
Do you consider that a well-trained external auditor would be doing good forensic work? Do you consider that a good forensic accountant would be properly serve as an internal auditor in the...
-
Watchdog to monitor soaring cost of flights as airlines keep supply low By Mark Saunokonoko, Senior Journalist, Dec 6, 2022 Australia's consumer watchdog has warnedairlinesthat the price of their...

Study smarter with the SolutionInn App


