Each of the following valence-shell configurations is possible for a neutral atom of a certain element. What
Question:
Each of the following valence-shell configurations is possible for a neutral atom of a certain element. What is the element and which configuration represents the ground state?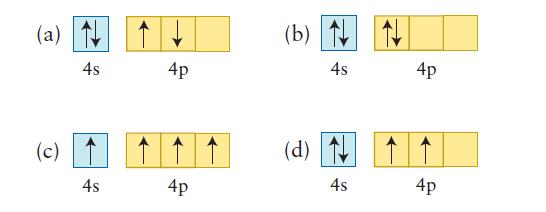
Transcribed Image Text:
(a) N 4s (c) 4s 4p 4p (b) N 4s (d) 4s 4p 4p
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The image youve provided shows four different electron configurations for the valence shells outermost energy levels of an atom Each configuration has ...View the full answer

Answered By

Jacob Festus
I am a professional Statistician and Project Research writer. I am looking forward to getting mostly statistical work including data management that is analysis, data entry using all the statistical software’s such as R Gui, R Studio, SPSS, STATA, and excel. I also have excellent knowledge of research and essay writing. I have previously worked in other Freelancing sites such as Uvocorp, Essay shark, Bluecorp and finally, decided to join the solution inn team to continue with my explicit work of helping dear clients and students achieve their Academic dreams. I deliver, quality and exceptional projects on time and capable of working under high pressure.
4.90+
1251+ Reviews
2842+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
1 . What are characteristics of a bond and what is the purpose of it ? 2 . What determines the Bond Price? 3 . What is the relationship between Price of a Bond and its return? What if the Price of a...
-
1) Match each of the following electron configurations to the correct atom. 1s 2 2s 2 2p 2 1s 2 2s 2 2p 6 3s 1 1s 2 2s 2 2p 6 3s 2 3p 5 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 3 1s 2 2s 2 2p 6 3s 2 3p 6 4s...
-
The beam shown below is supported by a pin at A and a roller at B. The weight of the beam is 12 kN and a 15 kN force is applied 3 m to the right of A as shown. Determine the reaction forces at A and...
-
The cylindrical plate is subjected to the three cable forces which are concurrent at point D. Express each force which the cables exert on the plate as a Cartesian vector, and determine the magnitude...
-
An astronomer finds a new absorption line with = 164.1 nm in the ultraviolet region of the Suns continuous spectrum. He attributes the line to hydrogens Lyman series. Is he right? Justify your...
-
A current-carrying wire has been bent into the form shown in Figure P28.74, with a half-circle of radius \(R_{1}\) lying in the \(x y\) plane connected via two straight segments to a half-circle of...
-
The Wendt Corporation had $10.5 million of taxable income. a. What is the companys federal income tax bill for the year? b. Assume the firm receives an additional $1 million of interest income from...
-
A 45-year-old man presents to the Emergency Department with a fever, widespread rash, and confusion. His only past history of note was Sydenham's chorea and a number of recent dental extractions....
-
The wavefunction for a particle in a one- dimensional box is given in Eq. 2. Does the probability of finding the particle in the left-hand one-third of the box depend on n ? If so, find the...
-
When an electron beam strikes a block of copper, x-rays with a frequency of 1.2 * 10 17 Hz are emitted. How much energy is emitted at this wavelength by (a) An excited copper atom when it generates...
-
Which modifiers are implicitly implied for interface variables? Select the three correct answers. (a) public (b) protected (c) private (d) default (e) abstract (f) static (g) final
-
Are investors in the money market best characterized as having a strong appetite for risk or being highly risk averse? What evidence would you use to support your answer?
-
What is the duration of a threeyear Treasury note with a coupon rate of 3 percent if the note is selling for par? What is its duration if the current yield to maturity is 10 percent? (Assume only one...
-
In what respect does a privately placed bond resemble a loan made by a commercial bank? Why do firms with access to the public bond market sometimes issue privately placed bonds under Rule 144a?
-
Describe the steps by which a change in the central banks policy interest rate affect the array of short-term interest rates.
-
Look for recent stories in the financial press about private equity funds or hedge funds. Explain the transactions that are being discussed and how this relates to the basic objective of the fund.
-
Grinders company issued the following statement of cash flows for 2011. (a) Compute free cash flow for Grinders Company. (b) Explain why free cash flow often provides better information than Net cash...
-
For the following exercises, find the area of the triangle. Round to the nearest hundredth. 22 50 36
-
You travel to a distant, cold planet where the ammonia flows like water. In fact, the inhabitants of this planet use ammonia (an abundant liquid on their planet) much as earthlings use water. Ammonia...
-
Provide a reasonable estimate for the number of atoms in a 150-lb adult human. Use the information given in Table 18.2. Table 18.2 Trace Elements Major Elemens Mass Percent in alphabetical order)...
-
Lead forms compounds in the 12 and 14 oxidation states. All lead(II) halides are known (and are known to be ionic). Only PbF4 and PbCl4 are known among the possible lead(IV) halides. Presumably lead...
-
Five Measures of Solvency or Profitability The balance sheet for Bearing Industries Inc. at the end of the current fiscal year indicated the following: Bonds payable, 6% Preferred $10 stock, $50 par...
-
Deacon Company is a merchandising company that is preparing a budget for the three-month period ended June 30th. The following Information is available Deacon Company Balance Sheet March 31 Assets...
-
Support department cost allocation-direct method Becker Tabletops has two support departments (Janitorial and Cafeteria) and two production departments (Cutting and Assembly). Relevant details for...

Study smarter with the SolutionInn App


