Ethylenediaminetetraacetate (EDTA 4- ) is used as a complexing agent in chemical analysis with the structure shown
Question:
Ethylenediaminetetraacetate (EDTA4-) is used as a complexing agent in chemical analysis with the structure shown in Fig. 19.8. Solutions of EDTA4- are used to treat heavy metal poisoning by removing the heavy metal in the form of a soluble complex ion. The complex ion virtually prevents the heavy metal ions from reacting with biochemical systems. The reaction of EDTA4- with Pb2+ is
Fig. 19.8
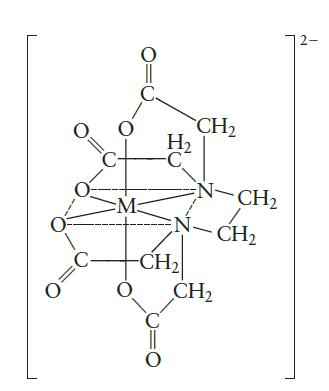
Transcribed Image Text:
O O M C H₂ -C CH₂ N-CH₂ 1 EN-CH₂ -CH₂ O CH₂ C O 2-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
The reaction of EDTA4 with Pb2 is shown below EDTA4 Pb2 EDTA4Pb2 I...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Ethylenediamineterraacetate (EDTA4-) is used as a complexing agent in chemical analysis with the structure show in. The trans isomer and its mior image are identical. They are not isomers of each...
-
A lens with a certain power is used as a simple magnifier. If the power of the lens is doubled, does the angular magnification increase or decrease?
-
A wooden board AB, which is used as a temporary prop to support a small roof, exerts at point A of the roof a 228-N force directed along BA. Determine the moment about C of that force. 0.12 m D. 18m
-
In figure, one end of a uniform beam weighing 20 x V3N is attached to a wall with a hinge. The other end is supported by a wire connected to the wall as shown. If the tension in the wire is a x 10N,...
-
Thirty-eight nurses working in the San Francisco area were surveyed concerning their opinions of managed health care. Identify the population and the sample.
-
The 0.8-kg particle is attached to the system of two light rigid bars, all of which move in a vertical plane. The spring is compressed an amount b/2 when = 0, and the length b = 0.30 m. The system...
-
Quilts R Us (QRU) is considering investing in a new patterning attachment with the cash flow profile shown in the table below. QRU's MARR is 13.5 percent/year. a. What is this investment's external...
-
The following information is available for Park Valley Spa for July 2016: The following is a list of checks and deposits recorded on the books of the Park Valley Spa for July 2016: Other Information...
-
Q6. Draw parallel realization of IIR by using the transfer function given by H(z) 3 (2 z+5Z+4)/ (2z+1) (z+2)
-
1. Using the post-acquisition integration matrix, Figure 11.3, compare Googles early style of acquisition management with the integration of Nest. 2. How has Google managed to be successful in...
-
Consider the following data: b. Based on your answer to part a, which is the stronger oxidizing agent, Co 3+ or Co(en) 3 3+ ? c. Use the crystal field model to rationalize the result in part b. Co+ +...
-
Nickel can be purified by producing the volatile compound nickel tetracarbonyl [Ni(CO) 4 ]. Nickel is the only metal that reacts directly with CO at room temperature. What is the oxidation state of...
-
Suppose the (quoted) yield on each of the six STRIPS increases by 0.05 percent. Calculate the percentage change in price for the one-year, three-year, and six-year STRIPS. Which one has the largest...
-
Sarah had the following income in 2023: Wages $4,000 Gifts from friend $2,000 How much can she contribute to a Roth IRA for tax year 2023?
-
Daisy Inc. had a fire destroy its warehouse on November 1, 2024. The following information was obtained from their accounting records: Description Amount Inventory (1/1/24) $ 20,000 Net Sales (1/1/24...
-
Trillium, Inc. reports the following information for April: Alpha Beta Units sold 5 comma 000 units 710 units Sales price per unit $ 410 $ 710 Variable manufacturing cost per unit 260 510 Sales...
-
Executive Services, Inc. has two business segments: Commercial and Residential. The following data have been provided for the year ending December 31, 20XX. Commercial Residential Service Revenue $ 7...
-
A project will cost $56,000 today and is forecasted to earn an inflow of $6,000 during the first year, followed by 8 annual cash flows of $12,000 thereafter. Cost of capital is 9.8% . What is this...
-
Toby's Trucking Company determined that the distance traveled per truck per year is normally distributed, with a mean of 50 thousand miles and a standard deviation of 12 thousand miles. 1. What...
-
a. What is the cost of borrowing if Amarjit borrows $28 500 and repays it over a four-year period? b. How many shares of each stock would he get if he used the $28 500 and invested equally in all...
-
A scientist is investigating a sample of 1.00 mol He(g) in a large cylinder at 200. K. Calculate by what factor each of the following variables would change the pressure of the gas and its root mean...
-
A sample of ethene gas, C 2 H 4 , of volume 1.00 L at 1.00 atm and 298 K is burned in 4.00 L of oxygen gas to form carbon dioxide gas and liquid water at the same pressure and temperature. Ignore the...
-
Describe the structures of a silicate in which the silicate tetrahedra share (a) One O atom; (b) Two O atoms.
-
Use the following probability distribution to answer the following questions. I P(x) 3 0.13 7 0.09 14. 0.12 15 0.14 20 0.52 Calculate the mean, variance, and standard deviation of the distribution....
-
For the figure on the right, express the fractional part represented by the shaded area as a percent. The shaded area is s% (Type an integer, proper fraction, or mixed number.)
-
A manufacturer sells video games with the following cost and revenue functions (in dollars), where x is the number of games sold. Determine the interval(s) on which the profit function is increasing....

Study smarter with the SolutionInn App


