For the exercise in this topic, base your answers on the potentials listed in Table 6M.1 or
Question:
For the exercise in this topic, base your answers on the potentials listed in Table 6M.1 or Appendix 2B, with the exception of the reduction and oxidation of water at pH = 7: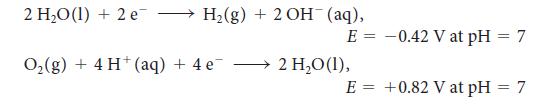 Suppose that 2.69 g of a silver salt (AgX) is dissolved in 550 mL of water. With a current of 3.5 A, 395.0 s was needed to plate out all the silver.
Suppose that 2.69 g of a silver salt (AgX) is dissolved in 550 mL of water. With a current of 3.5 A, 395.0 s was needed to plate out all the silver.
(a) What is the mass percentage of silver in the salt?
(b) What is the formula of the salt?
Transcribed Image Text:
2 H₂O(1) + 2 e → H₂(g) + 2 OH¯ (aq), E = O₂(g) + 4H+ (aq) + 4e¯ 2 H₂O (1), E = -0.42 V at pH = 7 +0.82 V at pH = 7
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
a ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
For the exercise in this topic, base your answers on the potentials listed in Table 6M.1 or Appendix 2B, with the exception of the reduction and oxidation of water at pH = 7: A sample of manganese of...
-
For the exercise in this topic, base your answers on the potentials listed in Table 6M.1 or Appendix 2B, with the exception of the reduction and oxidation of water at pH = 7: (a) How much time is...
-
For the exercise in this topic, base your answers on the potentials listed in Table 6M.1 or Appendix 2B, with the exception of the reduction and oxidation of water at pH = 7: Thomas Edison was faced...
-
- The first stream is $450 per year for 9 years and begins 5 years from today - The second stream begins 7 years from today with the first cash flow being $400, and with each successive cash flow...
-
Strategic analysis of operating income. Halsey Company sells womens clothing. Halseys strategy is to offer a wide selection of clothes and excellent customer service and to charge a premium price....
-
On January 1, 2011, Bradley Recreational Products issued $100,000, 9%, four-year bonds. Interest is paid semiannually on June 30 and December 31. The bonds were issued at $96,768 to yield an annual...
-
Visit the website of the marketing research company GMI (http://www.gmi-mr.com/) and write a brief report on the methods they use to ensure effective data processing for their clients.
-
You are considering investing in a company that cultivates abalone for sale to local restaurants. Use the following information: Sales price per abalone ............. = $ 35 Variable costs per...
-
Question -The US Trade Deficit - Is it Good or Bad for the US Economy? If so what's the reason?
-
Use the thermodynamic data in Appendix 2A to calculate the acidity constant of HF(aq). 2A THERMODYNAMIC DATA AT 25 C Inorganic Substances Substance Aluminum Al(s) Al+ (aq) AI(OH)3(s) AlO3(s) AICI,...
-
The pH of 0.50 m HBrO(aq) is 4.50. Calculate the change in pH when 5.10 g of sodium hypobromite is added to 100. mL of the solution. Ignore any change in volume.
-
Your one-story house has a floor area of \(2.0 \times 10^{2} \mathrm{~m}^{2}\) and 3. 0-m-high ceilings. (a) On a hot day, you leave home with all the windows closed and the air-conditioning on. A...
-
Discuss the components of the pension expense. In your opinion, is it reasonable to exclude some components of the change in the unfunded obligation from earnings? Is the calculation of the pension...
-
Suppose $X_{1}$ and $X_{2}$ are jointly normal positions with parameters $\mu_{1}, \mu_{2}, \sigma_{1}, \sigma_{2}, \sigma_{12}$. Show that \[\operatorname{VaR}_{h}\left(X_{1}+X_{2} ight) \leq...
-
A division of ABBOX Corporation has developed the concept of a new product. Production of the product would require \(\$ 10\) million in initial capital expenditure. It is anticipated that 1 million...
-
Nancy Smith says she is uncomfortable making the assumption that H&Ms dividend payout will vary from year to year. If she makes a constant dividend payout assumption, what changes does she have to...
-
What would be the value of Sanofis R&D asset at the end of fiscal years 2016 and 2017 if the average expected life of its R&D investments is only three years?
-
Kendle and Watson, a CPA firm, has provided audit and tax services to businesses in the London area for over 50 years. Recently, the firm decided to use ABM and activity-based costing to assign its...
-
You are planning to purchase your first home five years from today. The required down payment will be $50,000. You currently have $20,000. but you plan to contribute $500 each quarter to a special...
-
Consider the reaction FeO(s) + CO(g) Fe(s) + CO 2 (g) for which K P is found to have the following values: a. Calculate ÎG o R , ÎS o R , and ÎHR???? for this reaction at...
-
If K P is independent of pressure, why does the degree of dissociation in the reaction Cl 2 (g) 2Cl(g) depend on pressure?
-
How does the total number of moles in the reaction system change as T increases? H 2 (g) + Cl 2 (g) 2HCl(g) at equilibrium. Assume ideal gas behavior.
-
Credit cards can be powerful financial tools when used responsibly. They offer convenience, flexibility, and various benefits such as rewards programs, cashback offers, and fraud protection. However,...
-
Marc Corporation recently released zero-coupon bonds with a face value of $1,000. The bond has a duration of 22 years and a yield to maturity of 7.78 percent, with annual compounding. What is the...
-
15 years ago, Tom Inc issued zero-coupon bonds with a maturity date of 30 years and a face value of $1,000. Presently, the bond exhibits a semi-annually compounded yield to maturity of 11.58 percent....

Study smarter with the SolutionInn App


