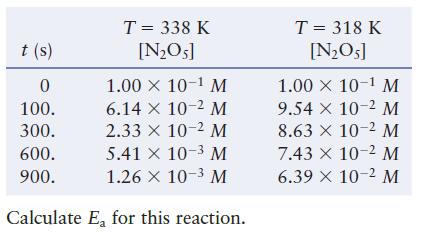
For the reaction 2NO5(g) 4NO2(g) + O(g)
Question:
For the reaction![]()
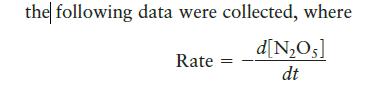
Transcribed Image Text:
2N₂O5(g) 4NO2(g) + O₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
The given reaction is represented below 2NO g NO g O g 1 Taking NO at 0 and 100 secon...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For the endothermic reaction the following represents a reaction container at two different temperatures. Which one (I or II) is at the lower temperature? AB(g) A(g) B(g),
-
For the following reaction profiles, indicate The positions of reactants and products. The activation energy. ÎE for the reaction. The second reaction profile is representative of a reaction...
-
For each of the following reaction coordinate diagrams, tell whether the structure of the transition state will more closely resemble the structure of the reactants or the structure of the products:...
-
Suppose a town concludes that it costs on average $30.00 per household to manage the disposal of the waste generated by households each year. It is debating two strategies for funding this cost: (1)...
-
A vending machine dispenses coffee into an eight-ounce cup. The amounts of coffee dispensed into the cup are normally distributed, with a standard deviation of 0.03 ounce. You can allow the cup to...
-
Name all of the alcohols that have the formula C 4 H 10 O. How many ethers have the formula C 4 H 10 O?
-
An unbalanced flywheel shows an amplitude of \(0.165 \mathrm{~mm}\) and a phase angle of \(15^{\circ}\) clockwise from the phase mark. When a trial weight of magnitude \(50 \mathrm{~g}\) is added at...
-
Columbus Limited is trying to determine the value of its ending inventory at February 28, 2014, the company's year-end. The accountant counted everything that was in the warehouse as of February 28,...
-
How does the Internet facilitate piracy? How might we explain the high levels of involvement in such practices by young people? Whose interests does the criminalization of copying serve? Is there a...
-
A rower wants to row her kayak across a channel that is 1400 ft wide and land at a point 800 ft upstream from her starting point. She can row (in still water) at 7 ft/s and the current in the channel...
-
Which of the changes listed below would affect the value of the rate constant k? a. Increasing the partial pressure of oxygen gas. b. Changing the temperature. c. Using an appropriate catalyst. - For...
-
For the reaction A + B C, explain at least two ways in which the rate law could be zero order in chemical A.
-
In Exercises find the indefinite integral. [(e-18 (et sin ti + et cos tj) dt
-
What is the difference between direct and indirect costs?
-
What are some of the budget types used within health services organizations?
-
What are the advantages of changing from a poor cost driver to a better one?
-
Why are planning and budgeting so important to an organizations success?
-
What are the two properties of an effective cost driver?
-
When examining a human pedigree, what features do you look for to distinguish between X-linked recessive inheritance and autosomal recessive inheritance? How would you distinguish X-linked dominant...
-
Perform the indicated operations. In designing a cam for a fire engine pump, the expression is used. Simplify this expression. (3) (3 4 32
-
Structural isomers are compounds that have the same chemical formula but the atoms are bonded together differently giving different compounds. Consider the two structural isomers having the formula...
-
The sp2 hybrid atomic orbitals have the following general form: where Ïs, Ïpx, and Ïpy represent orthonormal (normalized and orthogonalized) atomic orbitals. Calculate the values of A...
-
Experimental values for the temperature dependence of the rate constant for the gas-phase reaction NO(g) + O3(g) NO2(g) + O2(g) are as follows: Make the appropriate graph using these data, and...
-
With a one day sale on Christmas Day and same day return policy in full effect, people filled the stores of Macy's to shop and return. The total revenue sales numbers were 2M with $150,000 in...
-
An ethical organizational culture creates an environment in which to structure behavior that is then evaluated by stakeholders. The key elements of an organizational culture include all of the...
-
Identify and discuss the seven (7) key dimensions of organizational culture Describe what ZenDesk's organizational culture looks like Discuss how ZenDesk uses its corporate culture to attain...

Study smarter with the SolutionInn App


