Given the following data: calculate DH for the reaction CH(g) + O(g) C(s) + O(g) H(g) +
Question:
Given the following data:
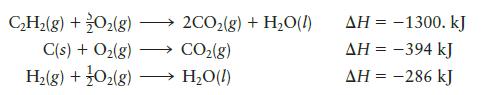
calculate DH for the reaction
![]()
Transcribed Image Text:
C₂H₂(g) + O₂(g) C(s) + O₂(g) H₂(g) + O₂(g) - →2CO₂(g) + H₂O(l) CO₂(g) → H₂O(l) AH = -1300. kJ AH = -394 kJ AH = -286 kJ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
Given the following data Eq1 C2H2 g 32O2g CO2g H2O l H...View the full answer

Answered By

Godswill Okorie
M.Sc chemistry specialization in organic chemistry, B.ed .I am having industry experience of seven years working with Ranbaxy nd Shimadzu analytical India by working as an application chemistry.I am having good practical experience on chromatography techniques,which later helped me in my teaching.I worked as PGT chemistry teacher with KV and APS.
As a teacher I was able to achieve good results with my students.I used to take 11th and 12th chemistry and science to classes 7th ,8th and ninth. While teaching I used to guide students for various carrier opportunities.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Given the following data from three in de pen dent samples, use the 0.025 level in determining whether the population means could be the same. (Use data file XR12092.) 3-48377 11212 701536 21121 2...
-
Given the following data on inputs and outputs at a work center, determine the cumulative deviation and the backlog for each time period. The beginning backlog is7. PERIOD Input Planned2 Actua2 200...
-
Given the following data from group 1 and group 2: a. Combine the two datasets in ranked order. Compute the sum of the ranks for group 1, R1. Compute the sum of the ranks for group 2, R2. b. Compute...
-
Find the magnitude and direction of the electric field strength at the point P due to the point charges at A and B as shown in the Figure 1. (k = 9 x 10 Nm/C) +2 C 10 cm A 10 cm Figure 1 B -8 C (5...
-
Suppose that a 2 test of independence at the level of significance 0.005 is to be applied to the elements of a 2 2 contingency table containing 2n observations, and that for some (0, 1) the data...
-
In 2016, Tom Morello established an S Corporation, RATM, to purchase a small apartment building in Cocoa Beach, FL. Tom borrowed $500,000 and contributed the same to RATM as a loan. From 2016 to...
-
Jack DeCoster owned Quality Egg, LLC, an Iowa egg production company. Jacks son, Peter DeCoster, served as the companys chief operating officer. Jack also owned and operated several egg production...
-
BusServ.com Corporation provides business-to-business services on the Internet. Data concerning the most recent year appear below: Sales . . . . . . . . . . . . . . . . . . . . . . . $8,000,000 Net...
-
A small electric immersion heater is used to heat 87 g of water for a cup of instant coffee. The heater is labeled "120 watts" (it converts electrical energy to thermal energy at this rate)....
-
Please review the State of West Virginia v. Keith O. Peoples case and respond to the following challenges: 1. The charge against Peoples was fraud, a criminal offense. Compare and contrast a criminal...
-
Given the following data: calculate DH for the reaction 2CIF(g) + O(g) 2CIF3(g) + 202(8) 2F(g) + O(g) ClO(g) + FO(g) ClO(g) + 3FO(g) 2FO(g) = 167.4 kJ = 341.4 kJ AH = -43.4 kJ
-
Given the following data: calculate DH for the reaction On the basis of enthalpy change, is this a useful reaction for the synthesis of ammonia? (g) + N (g) = 92 kJ (g) = 484 kJ
-
Carbon dioxide (CO 2 ) is heated in a constant-pressure process from 15 C and 101.3 kPa to 86 C. Determine, per unit mass, the changes in: (a) Enthalpy (b) Internal energy (c) Volume All in SI units....
-
Write a 300-500 word, double-spaced paper briefly describing the situation and critiquing the various aspects of the speaker's message and/or delivery - just as we have been doing in class. Also ,...
-
5. Write an assembly code for the following c statements: c=a +b*a; d = c +(b-a); using i. (1,3) GPR format ii. Stack architecture
-
Think about the background of your audience (who you communicate with on a weekly basis): how old are they? what kind of education do they have? what kind of families do they come from? where do they...
-
Assistance with this problem please. What is the formula for calculating Marginal cost? Continue with the table and complete the calculation for Marginal Cost: Bags/ Participants 0 100 200 300 400...
-
The Avengers You are a recruiting officer for S.H.I.E.L.D. Nick Fury, after consultation with Captain America, has made the decision to expand the core lineup of the Avengers. They want to add only...
-
Consider two identical buildings: one in Los Angeles, California, where the atmospheric pressure is 101 kPa and the other in Denver, Colorado, where the atmospheric pressure is 83 kPa. Both buildings...
-
You work as an operations consultant for a textile company. Your client has a well-established distribution system in the US market. The company has hundreds of stores and four distribution centers....
-
Explain the photoelectric effect.
-
An atom moving at its root mean square velocity at 100oC has a wavelength of 2.31 10-11 m. Which atom is it?
-
The ground state ionization energy for the one electron ion Xm+ is 4.72 104 kJ/ mol. Identify X and m.
-
Given what you have learned this week, what are some of the nutritional differences you see between the two photos? After describing how they are different, please answer the following questions:...
-
A basketball is dropped from a given height (height), and on each bounce, it reaches a maximum height that is 55% of its previous height. Write a method that returns the height reached by the...
-
Individual Assignment 2 - The Bank Rate (12.5%) NOTE: TO ANSWER THIS ASSIGNMENT USE BANK OF CANADA WEBSITE. What is the main objective of monetary policy? What is meant by the Target for the...

Study smarter with the SolutionInn App


