Identify the reactions with K > 1 in the following list and, for each such reaction, identify
Question:
Identify the reactions with K > 1 in the following list and, for each such reaction, identify the oxidizing agent and calculate the standard cell potential.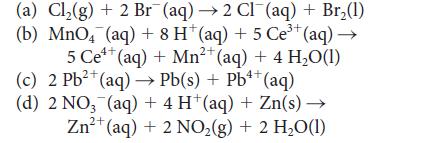
Transcribed Image Text:
(a) Cl₂(g) + 2 Br (aq) →2 Cl¯(aq) + Br₂(1) 3+ (b) MnO4 (aq) + 8 H* (aq) + 5 Ce³+ (aq) →→ 4+ 2+ 5 Ce¹+ (aq) + Mn²+ (aq) + 4 H₂O(1) 2+ 4+ (c) 2 Pb²+ (aq)→→ Pb(s) + Pb¹+ (aq) (d) 2 NO₂ (aq) + 4 H (aq) + Zn(s) → 2+ Zn²+ (aq) + 2 NO₂ (g) + 2 H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Clg 027 V ...View the full answer

Answered By

Geoffrey Isaboke
I am an industrious tutor with a 5-yr experience in professional academic writing. I have passion for History and Music and I have good knowledge in Economics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Identify the reactions with K > 1 among the following reactions and, for each such reaction, write balanced reduction and oxidation half-reactions. For those reactions, show that K > 1 by calculating...
-
Identify six (6) key areas of diversity from the following list and for each selected, give a brief description of what it means: cultural, disability, gender identity, generational, sexual identity,...
-
stock options are no longer as valuable and employees in startups are losing out to founders and early investors for all of these reasons except: there is higher employee turnover, less of a delay in...
-
A machine requires three hours to make a unit of Product A and nine hours to make a unit of Product B. Last month the machine operated for 957 hours, producing a total of 145 units. How many units of...
-
Analysis of growth, price-recovery, and productivity components (continuation of 13-23) Suppose that during 2009, the market for Merediths special-purpose machines grew by 3%. All increases in market...
-
Stanley-Morgan Industries adopted a defined benefit pension plan on April 12, 2011. The provisions of the plan were not made retroactive to prior years. A local bank, engaged as trustee for the plan...
-
What are the advantages of systematic sampling over simple random sampling?
-
Kate Petusky prepared Addison Controls' balance sheet and income statement for 2013. Before she could complete the statement of cash flows, she had to leave town to attend to a family emergency....
-
Suppose you borrow $260,000 at 5.25% for 30 years, monthly payments. You pay 2 discount points. A. Your APR on this loan is 5.625%. What amount of other financing fees did you pay? B. Suppose that...
-
Suppose that 25.0 mL of 0.10 m CH 3 COOH(aq) is titrated with 0.10 m NaOH(aq). (a)What is the initial pH of the 0.10 m CH 3 COOH(aq) solution? (b)What is the pH after the addition of 10.0 mL of 0.10...
-
Draw the Lewis structure or symbol for each of the following species and identify each one as a Lewis acid or Lewis base: (a) NH3; (b) BF3; (c) Ag; (d) F; (e) H.
-
Determine whether the following sentences focus on features or benefits; rewrite them as necessary to focus on benefits. 1. All-Cook skillets are coated with a durable, patented nonstick surface. 2....
-
What are the Scrum events, when do they take place and what is the purpose of each event?
-
What's the relationship between an epic, increment, sprint and tasks?
-
How can social media be used to conduct an experiment?
-
What does MoSCow mean and how is it used?
-
What is one way of measuring whether randomization has been effective?
-
The Grand Mogul is a 5-star hotel in Mumbai, India. The hotel has installed its own electrical generators to supplement the normal power supply and to ensure uninterrupted power to its demanding...
-
Show that gj concave AHUCQ Abadie For nonnegative variables, we have the following corollary.
-
In Chapter 10, we will see that an acetylide ion (formed by treatment of acetylene with a strong base) can serve as a nucleophile in an S N 2 reaction: This reaction provides a useful method for...
-
Predict the product(s) obtained when each of the following compounds is treated with chloromethane and aluminum trichloride. Some of the compounds might be unreactive. For those that are reactive,...
-
Predict the major product obtained when each of the following compounds is treated with bromine in the presence of iron tribromide. (a) Bromobenzene (b) Nitrobenzene (c) ortho-Xylene (d)...
-
A pension fund manager is considering three mutual funds. The first is a stock fund, the second is a long-term government and corporate bond fund, and the third is a T- bill money market fund that...
-
Using the 2019 link below, please answer each of the following questions. Your responses do not need to be in complete sentences. However, while you have access to the link above, you will have to...
-
The owner of the "Hamburgruesas" business purchased imported furniture worth $550,000 pesos. He put down $55,000 and the rest to be paid one year at 20% interest compounded monthly. Two months after...

Study smarter with the SolutionInn App


