In a gas-phase equilibrium mixture of H 2 , I 2 , and HI at 500. K,
Question:
In a gas-phase equilibrium mixture of H2, I2, and HI at 500. K, [HI] = 2.21 * 10–3 mol · L–1 and [I2] = 1.46 * 10–3 mol · L–1. Given the value of the equilibrium constant in Table 5G.2, calculate the equilibrium molar concentration of H2.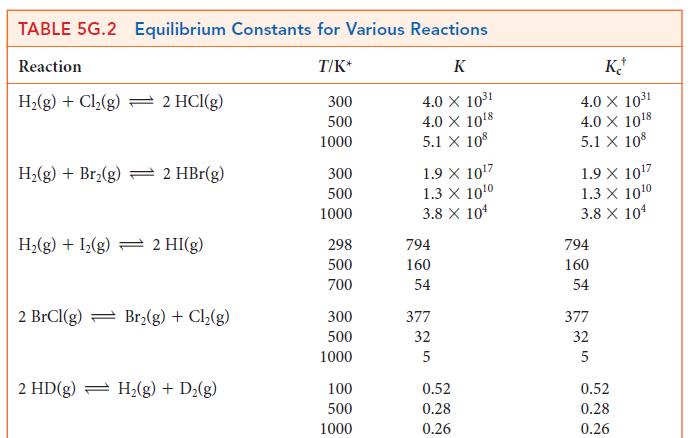
Transcribed Image Text:
TABLE 5G.2 Equilibrium Constants for Various Reactions Reaction T/K* K 4.0 X 10³1 4.0 X 10¹8 5.1 X 108 H₂(g) + Cl₂(g) = 2 HCl(g) H₂(g) + Br₂(g) + 2 HBr(g) H₂(g) + 1₂(g) 2 HI(g) 2 BrCl(g) Br₂(g) + Cl₂(g) 2 HD(g) H₂(g) + D₂(g) 300 500 1000 300 500 1000 298 500 700 300 500 1000 100 500 1000 1.9 X 10¹7 1.3 X 10¹0 3.8 X 10¹ 794 160 54 377 32 5 0.52 0.28 0.26 KJ 4.0 X 10³1 4.0 X 10¹8 5.1 X 108 1.9 X 10¹7 1.3 X 10¹0 3.8 X 104 794 160 54 377 32 5 0.52 0.28 0.26
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
H 2...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
The energy of activation for the decomposition of 2 mol of HI to H2 and I2 in the gas phase is 185kJ. The heat formation of HI to H2 and I2 is -5.65kJ/mol. Find the energy of activation for the...
-
Dinitrogen oxide, N 2 O, colloquially called laughing gas, was first used as an anesthetic in dentistry in 1844. Suppose that you are a chemist attempting to prepare N 2 O from N 2 and O 2 ; you...
-
An atom has a negative charge on it. What parameter is generally most important for determining its relative basicity? a. the type of atom that bears the negative charge b. the negative charge on the...
-
Perform the indicated operations and simplify the result. Leave your answer in factored form. X x - 3 x + 1 x2 + 5x 24
-
Two identical, 10-F parallel-plate capacitors are given equal charges of 100 C each and are then removed from the charging source. The charged capacitors are connected by a wire between their...
-
How do you think Isabelles understanding of theory and method changed in adopting the approach she chose? Isabelle was very thoughtful. She had a puzzled look on her face. The dilemma she had was one...
-
Integral balances can also be used for heat transfer in a turbulent-flow boundary layer if a form for the velocity profile is assumed. A common form is the 1/7th-power law:...
-
The management of Kauer Company has engaged you to assist in the preparation of year-end (December 31) financial statements. You are told that on November 30, the correct inventory level was 150,000...
-
What is the present value of the following future amount? $311,205 to be received 15 years from now, discounted back to the present at 5 percent, compounded annually.
-
Kenny Young has started his own business, Home and Away Inspections. He inspects property for buyers and sellers of real estate. Young rents office space and has a part- time assistant to answer the...
-
Hydrogen iodide, HI, is used as a reagent in organic chemistry to transform primary alcohols into alkyl iodides. Suppose you are a chemist using HI; you would need to understand the equilibrium...
-
You are now a chemical engineer in the process of designing a plant to separate hydrocarbons obtained from crude oil. You need to keep track of the composition of the mixtures you are dealing with,...
-
Write cos 4xcosx in terms of powers of cos x.
-
Consider a position consisting of a $100,000 investment in asset A and a $100,000 investment in asset B. Assume that the daily volatilities of both assets are 1% and that the coefficient of...
-
The engineering team at Manuels Manufacturing, Inc., is planning to purchase an enterprise resource planning (ERP) system. The software and installation from Vendor A costs \($380\),000 initially and...
-
A civil engineer is considering two pumps for an irrigation system. Pump A can be purchased for \($19\),400 with annual operating and maintenance expenses estimated to be \($7\),000. Alternatively,...
-
Some time ago a company entered into a forward contract to buy 1 million for $1.5 million. The contract now has 6 months to maturity. The daily volatility of a 6-month zero-coupon sterling bond (when...
-
(a). Using the list of transactions at question A5.5 prepare a spreadsheet similar to that presented in Table5.3. (b). Show that the spreadsheet totals satisfy the accounting equation. Table 5.3...
-
Determine the break-even contribution margin ratio for a company with a fixed overhead of $92,000, revenues of $450,000, and a required level of profit of $45,000.
-
on 8 For the following set of lengths 130, 170, 160, 160, 150, 190 Third quartile is: et red d out of Select one: O a. 160 a question O b. 145 O c. 175 O d. 180
-
Predict the products for each of the following reactions. a. b. c. d. SH 1) NaOH Br 2) Br SNa
-
Propose a plausible synthesis for each of the following transformations. a. b. c. d. e. CN En
-
Ethylene glycol is one of the main components of automobile antifreeze. Using iodoethane as your starting material, show how you could prepare ethylene glycol.
-
Ball bearing rated for 3000 hours at 500 RPM with C10=8850N. If the design life is 810^8 revolutions at 1000 rpm, what is FD?
-
You are replacing an HVAC system in your building. The system is a steam system that consists of a boiler that feeds several air-handling units. The total heating load for the building is 3613...
-
3 kg of gas in a piston-cylinder assembly undergo a process during which the relationship between pressure and specific volume is pv^1.25 = constant. The process begins with p_1 = 300 kPa and V_1 =...

Study smarter with the SolutionInn App


