Name the following acids. A formula of Na2X. Which of the following statements is(are) true? Correct the
Question:
Name the following acids. 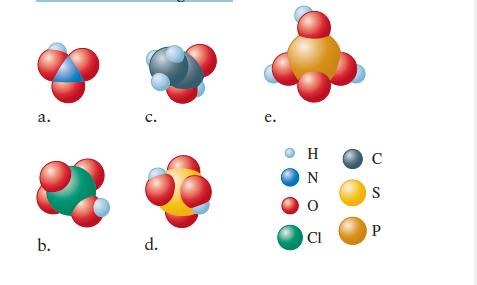
A formula of Na2X. Which of the following statements is(are) true? Correct the false statements.
a. The binary compound formed between X and fluorine will be a covalent compound.
b. The isotope of X contains 38 protons.
c. The isotope of X contains 41 neutrons.
d. The identity of X is strontium, Sr.
Transcribed Image Text:
a. b. C. d. e. H N CI C S P
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
a The statement is FALSE The binary compound formed betw...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Protons and neutrons (together called nucleons) are held together in the nucleus of an atom by a force called the strong force. At very small separations, the strong force between two nucleons is...
-
The only stable isotope of fluorine is fluorine-19. Predict possible modes of decay for fluorine-21, fluorine-18, and fluorine-17.
-
A strontium isotope has a half-life of 90 years. What is the continuous compound rate of decay? Use the radioactive decay model in Problem 33. Radioactive decay model Q = Q0ert where Q0 = amount of...
-
Which sentence below most strongly emphasizes the importance of the decision? (5 points) Over the next year, the direction of the company depends on the quick decision we must make. The direction of...
-
Three students A, B, and C are enrolled in the same class. Suppose that A attends class 30 percent of the time, B attends class 50 percent of the time, and C attends class 80 percent of the time. If...
-
Lilly Painting Company is considering whether to purchase a new spray paint machine that costs $4,000. The machine is expected to save labor, increasing net income by $600 per year. The effective...
-
Water flows over the sharp-crested weir shown in Fig. P10.86. The weir plate cross section consists of a semicircle and a rectangle. Plot a graph of the estimated flowrate, \(Q\), as a function of...
-
Cortez Chemical, Inc., requires 20,000 gallons of material annually; the cost of placing an order is $20, and the annual carrying cost per gallon is $5. Required: Determine the EOQ from potential...
-
The construction of a new coal mine will cost Yamada Co. Ltd. $430,000, with the expenditure occurring the next year. The investment will bring about cash flows of $200,000 annually over the...
-
The following activities and times (in minutes) were recorded for a press operator: removes completed widget from press (0.1), files rough edges on widget (0.4), blows filings from widget (0.3),...
-
The early alchemists used to do an experiment in which water was boiled for several days in a sealed glass container. Eventually, some solid residue would begin to appear in the bottom of the flask....
-
For each of the following sets of elements, label each as both noble gases, halogens, alkali metals, alkaline earth metals, or transition metals. a. Ti, Fe, Ag d. Ne, Kr, Xe b. Mg, Sr, Ba e. F, Br, I...
-
Electric baseboard heaters will be used to heat a room with a design heating load of 5050 Btu/hr. Identify the length of each of the following units (in feet) required to meet the load. a. Standard...
-
3. Suppose a firm uses the NPV decision rule. Considering the following cash flows, should the firm accept this project: Year 0 1 2 3 a. a required return of 11%? b. a required return of 25%? 4. What...
-
The Black Limo Company (BLC) purchased a limo on January 1 of Year 1. The limo cost $48,000. It had an expected useful life of 4 years and a $8,000 salvage value. Assuming BLC uses straight-line...
-
What cultural and political factors contribute to its popularity, and how does it reflect the intersection between reality and fantasy?
-
The current expected return on the ABC stock is 10%, the riskless rate of interest is 2%, and the return on the market portfolio is 8% and the standard deviation of returns on the market portfolio is...
-
Compute the present value of a perpetual bond that pays a monthly cash flow of $ 1 , 0 0 0 at an annual interest rate of 1 2 percent.
-
Compute the percents ionic character of the interatomic bonds for the following compounds: TiO2, ZnTe, CsCl, InSb, and MgCl2.
-
In Problem 8.43, determine the smallest value of for which the rod will not fall out of the pipe. IA -3 in.-
-
How long will it take to plate out each of the following with a current of 100.0 A? a. 1.0 kg of Al from aqueous Al 3+ b. 1.0 g of Ni from aqueous Ni 2+ c. 5.0 moles of Ag from aqueous Ag +
-
What mass of each of the following substances can be produced in 1.0 h with a current of 15 A? a. Co from aqueous Co 2+ c. I 2 from aqueous KI b. Hf from aqueous Hf 4+ d. Cr from molten CrO 3
-
Why is the electrolysis of molten salts much easier to predict in terms of what occurs at the anode and cathode than the electrolysis of aqueous dissolved salts?
-
Spade Millhone Detective Agency performs investigative work for a variety of clients. Recently, Alban Insurance Company asked Spade Millhone to investigate a series of suspicious claims for whiplash....
-
HomeWeb is a new real estate advertising company in Melbourne, Australia. The company has around 30 staff taking care of their main company website where real estate agents pay to post their...
-
Explain three (3) fundamentals of digital applications in a workplace What are the key digital applications that can be used in organisations? Explain three (3) key features of digital applications...

Study smarter with the SolutionInn App


