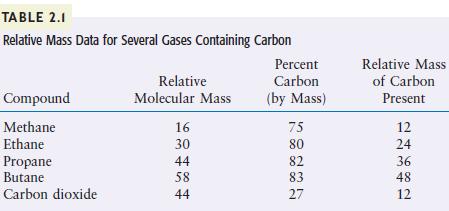
The first four compounds listed in Table 2.1 contain only carbon and hydrogen. Predict the formulas for
Question:
The first four compounds listed in Table 2.1 contain only carbon and hydrogen. Predict the formulas for these compounds.
Transcribed Image Text:
TABLE 2.1 Relative Mass Data for Several Gases Containing Carbon Compound Methane Ethane Propane Butane Carbon dioxide Relative Molecular Mass 16 30 44 58 44 Percent Carbon (by Mass) 75 80 82 83 27 Relative Mass of Carbon Present 243420 12 36 48 12
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Since the compounds contain only carbon and hydrogen the per cent hydrogen in each compoun...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Bioremediation is the use of microorganisms to degrade environmental pollutants. Many pollutants contain only carbon and hydrogen (oil being one example). The chemical reactions are complicated, but...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
THIRD AVENUE SOFTWARE HEALTH-CARE APP PROJECT This case is new for the ninth edition of Information Technology Project Management . The case provides an opportunity to apply agile and Scrum...
-
As an HR director for IMF Plastics Manufacturing, you receive a phone call about communicating changes made by the company to provide additional shifts.As soon as the call ends, you begin the process...
-
A counterweight D of mass 40 kg is attached to a cable that passes over a small pulley at A and that is attached to a support at B. Knowing that L = 15 m and h = 5 m, determine (a) The length of the...
-
For each of the independent transactions listed, indicate which of the revenue or contribution classifications apply by choosing one or more of the letters from the listed items in the right-hand...
-
Leicht Transfer & Storage provides warehousing services and often purchases pallets from Pallet Central. The companies followed a standard practice for documenting these transactions in which Pallet...
-
During the month of October 20--, The Pink Petal flower shop engaged in the following transactions: Oct. 1 Sold merchandise on account to Elizabeth Shoemaker, $1,000, plus tax of $50. Sale No. 222. 2...
-
explain diagram of ER model and relational model and attach their diagrams too Each cinema is identified by its name and has its residency at an address which consists of a street and city only....
-
Give the systematic name of each of the following compounds. a. CoBr 2 b. CaCl 2 c. Al 2 O 3 d. CrCl 3
-
Show that F = (3, 1, 2) is conservative. Then prove more generally that any constant vector field F = (a, b, c) is conservative.
-
Robert Harrity, the principal of Northeast Yachts, acted as an agent in the sale of a Bayliner boat owned by Malcolm Clark and sold to Margaret Kennedy. As Kennedy looked over the boat, Harrity...
-
Honda is selling a 2014 Accord Coupe Ex M for $30 056.90 including freight, PDI and all applicable fees. The lease payment of $382 is due at the beginning of each month. If the interest rate...
-
The target population for Mr, Prince's study is 5678 secondary school teachers, a size that seems so big against his financial capacity and time. Implying that Mr, Prince must deal with a statistical...
-
Consider a utility function given by U= x 1 1/2 x 2 3/2 . What is the MRS 2 1 ? Please provide explaination
-
What problems did Spyder face in 1995/1996 when Jacobs sought outside investors? EXPLAIN BRIEFLY
-
Describe some steps you took to investigate the USB image. You should have a well-thought-out approach for identifying evidence. Screenshots supporting your activities are required for full credit
-
The following selected data were taken from the financial statements of Preslar Inc. for December 31, 2012, 2011, and 2010: The 2012 net income was $435,750, and the 2011 net income was $453,750. No...
-
Research an article from an online source, such as The Economist, Wall Street Journal, Journal of Economic Perspectives, American Journal of Agricultural Economics, or another academic journal. The...
-
What is diafiltration? How does it differ from continuous feed-and-bleed ultrafiltration? Under what conditions is diafiltration used in conjunction with continuous feed-and-bleed ultrafiltration?
-
In Example 8.19, the % extraction of lysozyme is high (95.2%), but the % purity of the lysozyme in the total extracted proteins is low (15.1%). The Kremser plot of Figure 5.9 suggests that by...
-
What is continuous feed-and-bleed ultrafiltration? What are its limitations?
-
Interest versus dividend expense Michaels Corporation expects earnings before interest and taxes to be $40,000 for the current period. Assuming a flat ordinary tax rate of 21%, compute the firm's...
-
What managerial accounting tool did you find most useful and why?
-
A test requires that you answer first Part A and then either Part B or Part C. Part A consists of 7 true/false questions, Part B consists of 9 multiple choice questions with one correct answer out of...

Study smarter with the SolutionInn App


