The unit cell for a pure xenon fluoride compound is shown below. What is the formula of
Question:
The unit cell for a pure xenon fluoride compound is shown below. What is the formula of the compound? 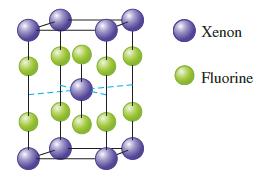
Transcribed Image Text:
Xenon Fluorine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
Answer The formula of the compound is XeF4 Explanation The formula of t...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The unit cell for a pure xenon fluoride compound is shown below. What is the formula of the compound? Xenon Fluorine
-
The unit cell for a pure xenon fluoride compound is shown below. What is the formula of the compound? Xenon Fluorine
-
Changing preferences can also affect changes in land use. In the United States, the proportion of the population in the 65-and-older age bracket is growing. What effects might this have on the...
-
For a sample of n = 64, find the probability of a sample mean being less than 24.3 when = 24 and = 1.25. The population mean and standard deviation are given. Find the indicated probability and...
-
Evaluate the expression. Assume x 0. a) (-6) 0 b) -(-6) 0
-
The following scenarios describe situations faced by hypothetical companies that currently have a centralized organization structure. As you review each of the scenarios, provide your opinion as to...
-
Winkler Furniture manufactures two different types of china cabinets: a French provincial model and a Danish Modern model. Each cabinet produced must go through three departments: carpentry,...
-
1. there are two different schools of thought on how to lower gasoline prices and reduce U.S. dependence on foreign oil. One is to increase supply of oil (drilling offshore), and the other is to...
-
Thompson Paint Company uses 60,000 gallons of pigment per year. The cost of ordering pigment is $200 per order, and the cost of carrying the pigment in inventory is $1 per gallon per year. The firm...
-
Consider the following melting point data: Compound NaCl MgCl AlCl3 SiCl4 PC13 SCl Cl mp (C) 801 708 190 -70 -91 -78 -101 Compound NaF mp (C) MgF 997 1396 AlF3 1040 -90 SiF4 PF5 SF6 F -94 -56-220
-
Use the diagram of the unit cell for the hexagonal closest packed structure in Fig. 16.14 to determine the net number of atoms in the hcp unit cell. Fig. 16.14 b a b 5 10 hep 12 3 Figure 16.16 The...
-
Helox Ltd manufactures a product that passes through two production processes. A quantity schedule for a recent month for the first process follows: Costs in the beginning work in progress inventory...
-
Give an example of a good or service that will have a negative income elasticity of demand but a low price elasticity of demand. Explain carefully why.
-
Assume that $200 is deposited today, two years from now, four years from now, six years from now, and eight years from now in a savings account which earns 10% annual interest, what is the balance at...
-
What are the primary uses of buckwheat, canola, grain sorghum, and sunflower?
-
9. If during 2011 the money supply increases by 6%, the inflation rate is 5%, and the growth of real GDP is 3%, what must have happened to the value of velocity during 2011?
-
Comparative balance sheets for 2018 and 2017 and a statement of income for 2018 are given below for Metagrobolize Industries. Additional information from the accounting records of Metagrobolize also...
-
The branch manager of a savings bank has recorded the number of errors of a particular type that each of 12 tellers has made during the past year. The results (stored in Teller) are as follows: a. Do...
-
In Exercises, find the equation of the tangent line at the given point on each curve. 2y 2 - x = 4; (16, 2)
-
Use Figs. 45 and 46 to answer the following questions. a. Would the bonding MO in HF place greater electron density near the H or the F atom? Why? b. Would the bonding MO have greater fluorine 2p...
-
The diatomic molecule OH exists in the gas phase. OH plays an important part in combustion reactions and is a reactive oxidizing agent in polluted air. The bond length and bond energy have been...
-
What is delocalized bonding, and what does it explain? Explain the delocalized bonding system in C6H6 (benzene) and SO2.
-
After 20 years of marriage, Ajani and Tanesha divorced. As per court instructions, effective January and for the full year, Ajani paid monthly spousal support to Tanesha in the amount of $450 and...
-
Evaluate (F(s)).
-
CODE Write output screen of the following program for each iteration. CONSOLE #include #include int main() { int ij,height=4; for(i=1;i

Study smarter with the SolutionInn App


