The values of K a for phenol and 2,4,6-trichlorophenol are 1.3 * 10 10 and 1.0 *
Question:
The values of Ka for phenol and 2,4,6-trichlorophenol are 1.3 * 10–10 and 1.0 * 10–6, respectively. Which is the stronger acid? Account for the difference in strength.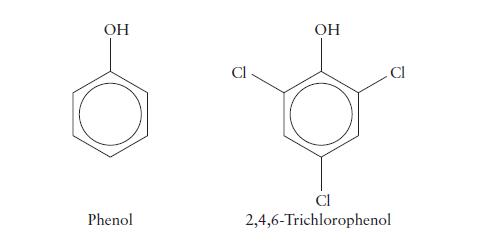
Transcribed Image Text:
OH Phenol OH Cl 2,4,6-Trichlorophenol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
246Tric...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
As a part of your research program on formic acid, you need to titrate a solution of formic acid with sodium hydroxide solution and want to know what to expect. Calculate the pH of (a) 0.100 m...
-
The amino acid glycine (H2N-CH2-COOH) can participate in the following equilibria in water: (a) Use the values of Ka and Kb to estimate the equilibrium constant for the intramolecular proton transfer...
-
Consider the acids in Table. Which acid would be the best choice for preparing a pH = 7.00 buffer? Explain how to make 1.0 L of this buffer. Table Values of Ka for Some Common Monoprotio Acids...
-
The coordinates of points A and B are given below: Easting Northing Height (meter) (meter) (meter) 41.676 66.446 225.973 127.066 31.063 185.401 Point A B What is the bearing of AB (from A to B)?...
-
Joint products and byproducts (continuation of 16-16). Quality Chicken is computing the ending inventory values for its July 31, 2009, balance sheet. ending inventory amounts on July 31 are 15 pounds...
-
The City Commission of Nashville has decided to build a botanical garden and picnic area in the heart of the city for the recreation of its citizens. The precedence table for all the activities...
-
Nuts and Cholesterol Several studies have been performed to examine the relationship between nut consumption and cholesterol levels. Here we consider two such studies. In Study 1, participants were...
-
The Chief Financial Officer of Eaton Medical Devices has determined that the firm's capital investment budget will be $5,000,000 for the upcoming year. Unfortunately, this amount is not sufficient to...
-
You create a new project roadmap that is visually appealing and contains all the information for the stakeholders to reference. However, this document took a lot of time to create and has caused some...
-
Write the chemical equations of the two proton transfer equilibria that demonstrate the amphiprotic character of (a) HCO 3 ; (b) HPO 4 2 . Identify the conjugate acidbase pairs in each case.
-
Calculate the molar solubility of silver bromide in 0.10 m KCN(aq).
-
Given the two probability distributions (a) Construct probability histograms. Which distribution has a larger spread? (b) Verify that both distributions have the same mean. (c) Compare the two...
-
Do you feel that daily fantasy leagues are gambling or games of skill? How do you feel the legislative and judicial branches of government will resolve this question in the future?
-
What is WAR (in the context of sport finance)? Conduct research to determine the ten most efficient players during the last MLB season. Then compare their salaries to determine which player offered...
-
If the federal inheritance tax is set at 40% of all assets above \($5\) million at the time of death, and a state's inheritance tax is set at 5% of all assets above \($1\) million at the time of...
-
According to an article on barrons.com, earnings increased for Starbucks in early 2019 partly because of lower variable costs due to streamlined operations. Draw a graph showing the effect of this...
-
Explain how the NBA's new luxury tax system operates. Research and explain an example where the new tax rates affected (or may have affected) a team's decision to sign players for its roster.
-
What are the three methods for allocating the costs of support activities?
-
Explain the regulation of the secretions of the small intestine.
-
Use the vapor pressures for hexane given in the following table to estimate the temperature and pressure of the triple point and also the enthalpies of fusion, vaporization, and sublimation. Phase T...
-
Show the paths n o p q and a b c d e f of the PVT phase diagram of Figure 8.15 in the PT phase diagram of Figure 8.4. Figure 8.4 Figure 8.15 Critical - point Liquid Solid Triple point Gas Tm...
-
Are the two PT phase diagrams below likely to be observed for a pure substance? If not, explain all features of the diagram that will not be observed. a. b. Liquid Solid Vapor Vapor Liquid Solid
-
What is National Savings Certificate? Explain please.
-
When recording direct materials usage, what does an unfavorable direct materials variance represents? Will this variance have a debit or credit balance?
-
A partnership consists of three partners: Monroe, Adams, and Madison who share profits equally. - Monroe loaned the partnership $ 1 0 , 0 0 0 & made a capital contribution of $ 2 0 , 0 0 0 . - Adams...

Study smarter with the SolutionInn App


