Use data from Tables 6C.1 and 6C.2 to place the following bases in order of increasing strength:
Question:
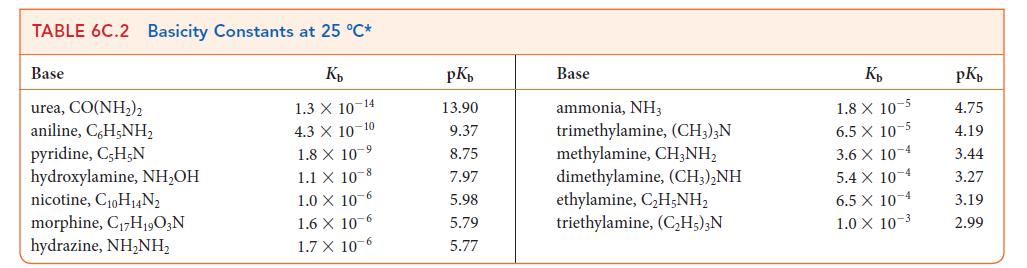
Use data from Tables 6C.1 and 6C.2 to place the following bases in order of increasing strength: F–, NH3, CH3CO2–, C5H5N (pyridine).
Transcribed Image Text:
TABLE 6C.1 Acidity Constants at 25 °C* K₂ 3.0 X 10-1 2.0 X 10-1 1.7 X 10-1 1.5 X 1.0 X 10-2 Acid trichloroacetic acid, CCI,COOH benzene sulfonic acid, C6H5SO3H iodic acid, HIO3 sulfurous acid, H₂SO3 chlorous acid, HClO₂ phosphoric acid, H3PO4 chloroacetic acid, CH₂ClCOOH lactic acid, CH₂CH(OH)COOH nitrous acid, HNO₂ hydrofluoric acid, HF 10 2 7.6 X 10-3 1.4 X 10-3 8.4 X 10 4 4.3 X 10-4 3.5 x 10-4 pKa 0.52 0.70 0.77 1.81 2.00 2.12 2.85 3.08 3.37 3.45 Acid formic acid, HCOOH benzoic acid, C,H₂COOH acetic acid, CH,COOH carbonic acid, H₂CO3 hypochlorous acid, HCIO hypobromous acid, HBrO boric acid, B(OH)3¹ hydrocyanic acid, HCN phenol, C,H,OH hypoiodous acid, HIO K₂ 1.8 X 10 4 6.5 X 10-5 1.8 X 10-5 4.3 X 107 3.0 X 108 2.0 × 10-⁹ 7.2 X 10-10 4.9 X 10-10 1.3 X 10-10 2.3 X 10-11 pKa 3.75 4.19 4.75 6.37 7.53 8.69 9.14 9.31 9.89 10.64
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
F 1400 345 ...View the full answer

Answered By

Lamya S
Highly creative, resourceful and dedicated High School Teacher with a good fluency in English (IELTS- 7.5 band scorer) and an excellent record of successful classroom presentations.
I have more than 2 years experience in tutoring students especially by using my note making strategies.
Especially adept at teaching methods of business functions and management through a positive, and flexible teaching style with the willingness to work beyond the call of duty.
Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with a colorful wing of future.
I do always believe that more than being a teacher who teaches students subjects,...i rather want to be a teacher who wants to teach students how to love learning..
Subjects i handle :
Business studies
Management studies
Operations Management
Organisational Behaviour
Change Management
Research Methodology
Strategy Management
Economics
Human Resource Management
Performance Management
Training
International Business
Business Ethics
Business Communication
Things you can expect from me :
- A clear cut answer
- A detailed conceptual way of explanation
- Simplified answer form of complex topics
- Diagrams and examples filled answers
4.90+
46+ Reviews
54+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Use data from Tables 6C.1 and 6C.2 to place the following bases in order of increasing strength: C 10 H 14 N 2 (nicotine), ClO 2 , (CH 3 ) 3 N, HSO 3 . TABLE 6C.1 Acidity Constants at 25 C* K 3.0 X...
-
Arrange the following bases in order of increasing strength on the basis of the pK a values of their conjugate acids, which are given in parentheses: (a) Aniline (4.63; see Exercise 6C.12); (b)...
-
Arrange the following bases in order of increasing strength on the basis of the pK a values of their conjugate acids, which are given in parentheses: (a) Ammonia (9.26); (b) Methylamine (10.56); (c)...
-
Christina is trying to save money in her bank account. She decides that she can deposit $1000 each month into the account. The account earns 2.3% interest each month. How much money will be in the...
-
Variance analysis, multiple products. Debbies Delight, Inc., operates a chain of cookie stores. Budgeted and actual operating data of its three Chicago stores for August 2009 are as follows: Debbies...
-
Construct a cause-and-effect diagram to reflect student dissatisfied with university registration process. Use the four Ms or create your own organizing scheme. Include at least 12 causes.
-
In each of the following situations, indicate the statistical analysis you would conduct and the appropriate test or test statistic that should be used. a. Consumer preferences for Coca-Cola were...
-
An automobile accident causes both the driver and passenger front airbags to deploy. (a) If the vehicle was traveling at a speed of 88.6 km/h and is now at rest, find the change in momentum for both...
-
2. For each of the following transactions, state two things: (1) which section of the Statement of Cash Flows (SCF) would this transaction be reported in; and (2) would it be ADDED or SUBTRACTED?...
-
Use the data in Appendix 2B to calculate E(U 4+ /U). 2B STANDARD POTENTIALS AT 25 C Potentials in Electrochemical Order Reduction half-reaction Strongly oxidizing H4XeO6 + 2 H+ 2 e XeO3 + 3 HO F+2 e...
-
Calculate the pH and pOH of each of the following aqueous solutions of a strong acid or base: (a) 0.0146 m HNO 3 (aq); (b) 0.11 m HCl(aq); (c) 0.0092 m Ba(OH) 2 (aq); (d) 2.00 mL of 0.175 m KOH(aq)...
-
A study of child care enrolled 1364 infants and followed them through their sixth year in school. Later, the researchers published an article in which they stated that the more time children spent in...
-
Cabana Cruise Line offers cruise ship service to various tropical locations throughout the globe. The company is considering the addition of a new cruise ship to its fleet in order to expand service...
-
You have a project with the following information: The total initial investment capital is 1,600 million VND for equipment. The installation of equipment and preparation for production will be...
-
Use the following data and write a VBA function to compute the price of a European Call and Put. You can use Black-Scholes-Merton Model to compute the price of the European Call and Put. Current Date...
-
2 Suppose that you estimate the following relationship using OLS: Y = Bo + BX + B Zi + ei However, the true data generating process for Y is: Y = Bo + BX + BZ + B3W + U where cov(X, u) = 0, cov(W, u)...
-
Consider the Solow growth model. Recall that upper-case variables denote aggregate variables and lower-case variables denote per worker (or per capita) variables. Capital is assumed to evolve...
-
Refer to Problem 15.32. Required: Complete steps 15 of Oranges process-costing report for June. What is Oranges cost of goods manufactured (COGM) for June and the cost of its work-in-process...
-
The MIT Sloan School of Management is one of the leading business schools in the U.S. The following table contains the tuition data for the masters program in the Sloan School of Management. a. Use...
-
Why is it not appropriate to use ionic radii from crystal structures to calculate G solvation of ions using the Born model?
-
Why do deviations from ideal behavior occur at lower concentrations for electrolyte solutions than for solutions in which the solute species are uncharged?
-
Why is the value for the dielectric constant for water in the solvation shell around ions less than that for bulk water?
-
The machinery has an estimated useful life of 15 years and a residual value of $11.050. The company uses the straight-line method of depreciation. At the end of 2022. Sheffield recorded depreciation...
-
Mission 1. What is a mission within a school? 2. Provide an example of a school mission 3. Please add how this mission is reflected (opening doors, manners etc)
-
# 1 a) Determine the representation of the following decimal numbers in the IEEE 32-bit format (-10.675) 10 b) Convert the following 32-bit machine number in IEEE 32-bit format to a decimal number....

Study smarter with the SolutionInn App


