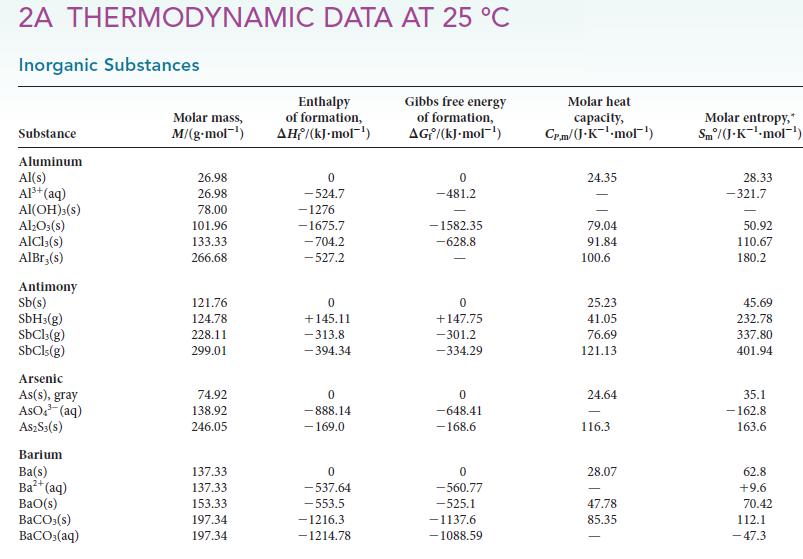
Use the data in Appendix 2A to calculate the standard reaction enthalpy for the reaction of pure
Question:
Use the data in Appendix 2A to calculate the standard reaction enthalpy for the reaction of pure nitric acid with hydrazine:![]()
Transcribed Image Text:
4 HNO3(1) + 5 N₂H4 (1) 7 N₂(g) + 12 H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
298...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Use the data in Appendix 2A to calculate the standard reaction enthalpy for the reaction of magnesium carbonate with hydrochloric acid: MgCO3 (s) + 2 HCI (aq) MgCl (aq) + HO(1) + CO(g)
-
Use standard enthalpies of formation from Appendix 2A to calculate the standard reaction enthalpy for each of the following reactions: (a) The final stage in the production of nitric acid: (b) The...
-
Use data in Table 4H.1 or Appendix 2A to calculate the standard entropy change for each of the following reactions at 25C. For each reaction, interpret the sign and magnitude of the reaction entropy....
-
A general ledger trial balance at June 30, 2011, for Millar City is as follows: Millar City uses a purchases basis in accounting for supplies. Open encumbrances are considered constrained by the...
-
Data for Sanchez Manufacturing are given in BE21-1. Supporting records show that (a) The Assembly Department used $24,000 of raw materials and $30,000 of the factory labor, and (b) The Finishing...
-
Solve the triangles with the given parts. a = 0.841, b = 0.965, A = 57.1
-
The fastest baseball pitchers can throw the \(0.145-\mathrm{kg}\) ball at speeds of about \(45 \mathrm{~m} / \mathrm{s}\). Ignoring air resistance, what height must the ball be dropped from to hit...
-
I am going to focus on the customers of my business and leave cost-allocation issues to my accountant. Do you agree with this comment by a division president? Why?
-
Fluorine Limited (FL), a manufacturer of ships, has entered into the following contracts during the year ended 31 December 2022: (i) On 1 January 2022, FL entered into a contract with Alpha Limited...
-
Calculate the standard entropy of vaporization of water at 85C, given that its standard entropy of vaporization at 100.C is 109.0 J K 1 mol 1 and the molar heat capacities at constant pressure of...
-
Use the following information to construct a heating curve for bromine from 27.2C to 70.0C. The molar heat capacity of liquid bromine is 75.69 J K 1 mol 1 and that of bromine vapor is 36.02 J K 1 ...
-
Refer to Exercise 61. Suppose the sample size here had been 20 instead of 49. Suppose, too, that the population standard deviation is unknown, but the sample has a standard deviation of 5.2 years. a....
-
A bailment includes a transfer of title. (True/False)
-
Which type of property does a bailment involve?
-
What does an incontestability clause provide?
-
Most bailments are created by agreement. (True/False)
-
What is an estray statute?
-
Why might the party favorites at a political convention sometimes be harder to elect than more moderate candidates?
-
Why should you not model a decision variable as a random variable with a probability distribution?
-
Define and give an example of each of the following. a. Addition polymer b. Condensation polymer c. Copolymer d. Homopolymer e. Polyester f. Polyamide
-
What is polystyrene? The following processes result in a stronger polystyrene polymer. Explain why in each case. a. Addition of catalyst to form syndioracric polystyrene b. Addition of 1,3-butadiene...
-
What monomer(s) must be used to produce the following polymers? a. b. c. d. e. f. Classify these polymers as condensation or addition polymers. Which are copolymers? CH-CH2-CH-CH-CH-CH O-CH2-CH2-C-o...
-
You are asked to recommend a new role to the CEO. The role's mission is to deepen the company's commitment to innovation and the use of advanced analytics. How should that role be defined? What title...
-
What tasks are specifically assigned to a unit by its higher headquarters during the second phase of the military decion making process?
-
Without performing any row operations, explain why the matrix does not have an inverse. 6 -2 4 -21 7 3

Study smarter with the SolutionInn App


