Use the information in Table 9C.1 to write the formula for each of the following coordination compounds:
Question:
Use the information in Table 9C.1 to write the formula for each of the following coordination compounds:
(a) Triamminediaquabromidocobalt(II) hydroxide
(b) Dichloridobisethylenediaminecobalt(III) bromide
(c) Sodium triamminetrichloridonickelate(II)
(d) Barium tris(oxalato)ferrate(III)
(e) Diaquadichloridoplatinum(IV) iodide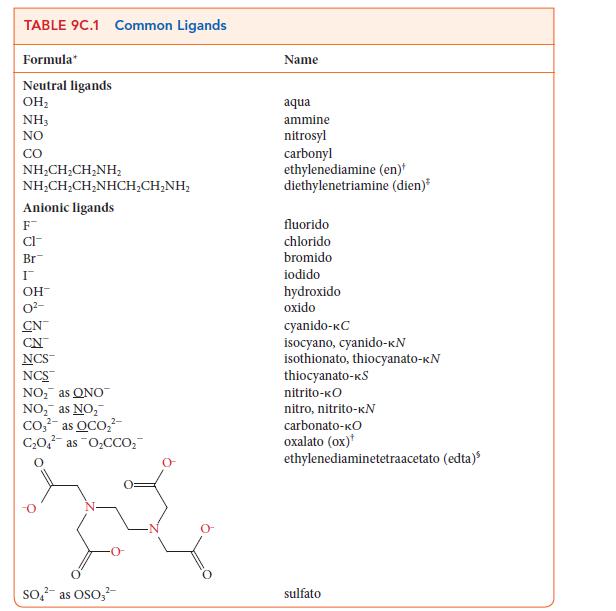
Transcribed Image Text:
TABLE 9C.1 Common Ligands Formula Neutral ligands OH₂ NH3 NO CO NHẠCH,CH_NH, NHẠCH,CH,NHCH,CHÍNH, Anionic ligands F CI- Br I OH- CN NCS™ NCS™ NO₂ as ONO NO₂ as NO₂ Co₂² as OCO₂² C₂O4 as O₂CCO₂ 2- SO² as OSO3² Name aqua ammine nitrosyl carbonyl ethylenediamine (en) diethylenetriamine (dien)* fluorido chlorido bromido iodido hydroxido oxido cyanido-KC isocyano, cyanido-kN isothionato, thiocyanato-KN thiocyanato-kS nitrito-KO nitro, nitrito-KN carbonato-kO oxalato (ox) ethylenediaminetetraacetato (edta) sulfato
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Solution a CoNH3...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Write the formula for each of the following compounds: a. Isobutyl chloride b. isopropyl bromide c. 2-chlorobutane d. tert-butyl iodide e. Propyl fluoride f. general formula for an alkyl bromide
-
Write the formula for each of the following compounds, being sure to use brackets to indicate the coordination sphere: (a) Hexaamminechromium (III) nitrate (b) Tetraamminecarbonatocobalt (III)...
-
Write a structural formula for each of the following compounds a. Sec-butyl tert-butyl ether b. Isoheptyl alcohol c. Sec-butylamine d. Neopentyl bromide e. 1,1-dimethylcyclohexane f....
-
What is true about business cycles? Multiple select question. They vary in duration and intensity. They follow an identical pattern. They are made up of alternating rises and declines. "Ups" are...
-
Dijkstra posed each of the following solutions as a potential software solution to the critical section problem and then explained why they failed [Dijkstra, 1968]. Provide your explanation about why...
-
At the end of 2013, Seiford Enterprises' Accounts Payable balance was $341,053; at the end of 2014, it was $160,094. Assume that the company purchased $1,951,523 of inventory during 2014 and that...
-
For each of the following situations, calculate the \(t\)-statistic \((t)\) : a. \(\mathrm{X}^{-}=.45 ; \mu=.52 ; s=.17 ; N=56\) b. \(\mathrm{X}^{-}=7.75 ; \mu=6 ; s=3.98 ; N=40\) c....
-
Steve Jackson (age 51) is a single taxpayer living at 3215 Pacific Dr., Del Mar, CA 92014. His Social Security number is 465-88-9415. In 2012, Steve's earnings and income tax withholding as the...
-
Macy Pharmacy has a project which has the following cash flows. Year 0 -$200,000 Year 1 = $50,000 Year 2 $100,000 Year 3 $150,000 Year 4 $40,000 = Year 5 $25,000 = The cost of capital is 10%. What is...
-
Draw an orbital energy-level diagram (like those in Figs. 9D.3 and 9D.5) showing the configuration of d-electrons on the metal ion in each of the following complexes: (a) [Co(NH 3 ) 6 ] 3+ ; (b)...
-
You are an engineer studying materials for computer hard-drive fabrication and need to predict the magnetic properties of the iron complexes you are investigating. Compare the magnetic properties of...
-
The Stahl Metalworks Corp. general ledger contained the following information at September 30, 2020: Cost of goods...
-
An electron is located at coordinates (-a, a); another electron is located at coordinates (-a, -a); a third electron is located at coordinates (a, -a), as shown in the figure below. Each electron has...
-
The U.S. Department of Transportation reported the accompanying data on the number of speeding-related crash fatalities during holiday periods for the years from 1994 to 2003. Holiday Period 1994...
-
When a force F acts on an object at some distance r from a point P, called the pivot point, it can cause the object to rotate rather than translate. The ability of a force to cause rotation is called...
-
The magnitude of E is given by 191 2 |E| = E = Ke E = ke where ke is Coulomb's constant and r is the distance, h, of P from 9. We have 191 2 r r1 I = Ke , Need Help? Read It h = (8.99 x 109 Nm/C2)-...
-
Consider the system of masses connected by light ropes that pass over massless and frictionless pulleys shown in the figure. If m = 1.0 kg and m = 2.5 kg, calculate (a) the accelerations of each...
-
Who should be in charge of food purchasing?
-
Why is homeostasis defined as the "relative constancy of the internal environments? Does negative feedback or positive feedback tend to promote homeostasis?
-
A set of 13 particles occupies states with energies of 0, 100, and 200 cm 1 . Calculate the total energy and number of microstates for the following configurations of energy: a. a 0 = 8, a 1 = 5, and...
-
For a set of non-degenerate levels with energy /k = 0,100, and 200. K, calculate the probability of occupying each state when T = 50, 500, and 5000.K. As the temperature continues to increase, the...
-
For two non-degenerate energy levels separated by an amount of energy /k = 500.K, at what temperature will the population in the higher-energy state be 1/2 that of the lower-energy state? What...
-
For companies that manufacture several products that use different proportions of manufacturing overhead resources, why does the use of activity-based costing provide more reliable product cost...
-
Night Shades, Incorporated, manufactures biotech sunglasses. The variable materials cost is $ 1 2 . 2 0 per unit, and the variable labor cost is $ 6 . 0 0 per unit. a . What is the variable cost per...
-
1. Discuss two preventative measures or internal controls that could have prevented accounting manager Nathan Meulerfrom stealing nearly $8.5 million in four years from his employer ING. 2. Research...

Study smarter with the SolutionInn App


