Using bond energies, estimate (Delta H) for the following reaction: CH3CHOH(aq) + HOCCH3(aq) CH3CHOCCH(aq) + HO(l)
Question:
Using bond energies, estimate \(\Delta H\) for the following reaction:
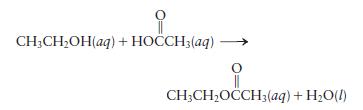
Transcribed Image Text:
CH3CHOH(aq) + HOCCH3(aq) CH3CHOCCH(aq) + HO(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Youve provided an image of a chemical reaction and asked to estimate the change in enthalpy Delta H for the reaction using bond energies The image dep...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When atoms of the hypothetical element X are placed together, they rapidly undergo reaction to form the X2 molecule: X(g) + X(g) X2(g) a. Would you predict that this reaction is exothermic or...
-
Estimate the value of Ho for the following reaction from bond energies (Table 9.5). H2(g) + Cl2(g) 2HCl(g). Is the reaction exothermic or endothermic? Note that the reaction involves the breaking of...
-
Create basic framework for your client using the completed ASI (I will provide the ASI document for students in class to complete). complete the following steps: Tell client's name and the...
-
Show that an emission tax and an absolute emission standard are equivalent instruments to regulate a polluting monopolist if and only if the standard is binding.
-
Let the initial position and speed of an over damped, non-driven oscillator be x0 and v0, respectively. (a) Show that the values of the amplitudes A1 and A2 in Equation 3.44 have the values A1 = 2x0...
-
Explain two ways that databases can be redesigned.
-
True or False: If \(P W>0\), then \(I R R>M A R R\).
-
Planning and control decisions. Conner Company makes and sells brooms and mops. It takes the following actions, not necessarily in the order given below. For each action (ae below,) state whether it...
-
Ruler Formula Bar Normal Page Break Page Custom Preview Layout Views Gridlines Headings Zoom 100% Zoom To Selection Window Macros Workbook Views Show Zoom Macros A A1 V A B D E F G H 4 Assume That...
-
Give a rationale for the octet rule and the duet rule for \(\mathrm{H}\) in terms of orbitals.
-
Write Lewis structures for \(\mathrm{CO}_{3}{ }^{2-}, \mathrm{HCO}_{3}{ }^{-}\), and \(\mathrm{H}_{2} \mathrm{CO}_{3}\). When acid is added to an aqueous solution containing carbonate or bicarbonate...
-
In the network in fig, the switch opens at t =0. Use Laplace transform to find I (t) for t>0.
-
Explain with the help of an example how external and internal forces can result in organizational change.
-
Explain how life cycle forces influenced competition at each stage of the life cycle?
-
Garret Company has provided the following selected information for the year ended December 31, 2022: Cash collected from customers was $784,000. Cash received from stockholders in exchange for common...
-
How can teachers facilitate the development of academic English for ELLs? What do teachers need to keep in mind as they raise the learning expectations for students who are not as confident in their...
-
Explain each step in the SMART method used for setting job performance standards and developing personal performance goals
-
Krylon Company purchases eight special tools annually from CO., Inc. The price of these tools has increased each year, reaching $100,000 per unit last year. Because the purchase price has increased...
-
Compare and contrast debt financing and equity financing as ways of starting a new business. Does one have an overall advantage over the other? What situation is more favorable to the use of debt...
-
Calculate the pH of the solution that results from mixing (a) 0.100 L of 0.050 m (CH 3 ) 2 NH(aq) with 0.280 L of 0.040 m (CH 3 ) 2 NH 2 Cl(aq); (b) 45.0 mL of 0.015 m (CH 3 ) 2 NH(aq) with 86.0 mL...
-
Morphine, C 17 H 19 O 3 N, is a potent painkiller. Suppose you are studying morphine and need to predict the pH of a morphine solution during a titration. Calculate the pH at the stoichiometric point...
-
Calculate the molar concentrations of H 2 SO 3 , HSO 3 , SO 3 2 , H 3 O + , and OH present in 0.125 m H 2 SO 3 (aq).
-
6) Bill and Ted want to go on an EXCELLENT ADVENTURE (vacation) in 4 years. If their savings account pays 5.75% compounded semi-annually and they are saving $250 per end of month, determine: (5...
-
Suppose you bought a three-year CD for $20,000 with an APR of 3.88% compounded quarterly. Determine the amount of interest you will have earned when the CD matures? Round your answer to the nearest...
-
Calculate the cumulative incidence, prevalence, all-cause mortality, cause-specific mortality, and case fatality using the below data. Year Sex Age Groups New Cases Existing cases Death among those...

Study smarter with the SolutionInn App


