Using the K a K a values given in Table 7.2, calculate the concentrations of all species
Question:
Using the Ka values given in Table 7.2, calculate the concentrations of all species present and the pH for each of the following.
a. 0.20MHOCl
b. 1.5MHOC6H5
c. 0.020MHF
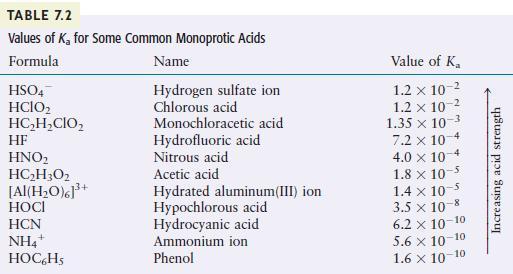
Transcribed Image Text:
TABLE 7.2 Values of K for Some Common Monoprotic Acids Formula Name Hydrogen sulfate ion Chlorous acid HSO4 HCIO HCHCIO HF HNO HCH3O2 [Al(HO)]+ HOCI HCN NH4+ HOC6H5 Monochloracetic acid Hydrofluoric acid Nitrous acid Acetic acid Hydrated aluminum(III) ion Hypochlorous acid Hydrocyanic acid Ammonium ion Phenol Value of K 1.2 x 10- 1.2 x 10-2 1.35 x 10-3 7.2 x 10-4 4.0 x 10 1.8 x 10-5 1.4 x 10- 3.5 x 10-8 6.2 10-10 5.6 10-10 1.6 10-10 Increasing acid strength
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The question involves calculating the concentrations of all species present and the pH for different solutions of monoprotic acids using given Ka valu...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the concentrations of all species in a 0.100 M H3PO4 solution.
-
The gamma distribution may be written in several different (but mathematically equivalent) forms. Excel uses the following form for the two-parameter gamma distribution in its functions GAMMADIST...
-
Write a short paper describing your current (or most recent) job and the compensation system in place at your current (or most recent) place of employment. Dont use the name of a company, just...
-
West State Furniture (WSF) manufactures desks and desk chairs using two departments within a single facility. The West Department produces the desks, and the State Department produces the chairs. WSF...
-
Determine the minimum multiplication of a telescope with diameter of objective D = 5.0 cm with which the resolving power of the objective is totally employed if the diameter of the eye's pupil is do...
-
With HTTP streaming, are the TCP receive buffer and the clients application buffer the same thing? If not, how do they interact?
-
A significant issue when a contract is breached is whether the cause of action is strictly in breach of contract or may be expanded to negligence and strict liability. The significance comes into...
-
Based on the following data, calculate the net income for the period and the balance in the account for Nuno Texeira, Capital after the closing entry has been posted. Cash . $ 12,000 Accounts...
-
Amajorlandscape contractor who has bid successfully onalarge-scale Boston beautification and urban greening project has offered to buy all 10,000 flowering dogwood trees ataprice of $28,000, payable...
-
Monochloroacetic acid \(\left(\mathrm{HC}_{2} \mathrm{H}_{2} \mathrm{ClO}_{2}ight)\) is a skin irritant that is used in "chemical peels" intended to remove the top layer of dead skin from the face...
-
List the major species present in \(0.250 \mathrm{M}\) solutions of each of the following acids, and then calculate the \(\mathrm{pH}\) for each. a. \(\mathrm{HBr}\) b. \(\mathrm{HClO}_{4}\) c....
-
Ms. GW, an unmarried taxpayer, had the following income items: Salary ..$33,400 Net income from a rental house .. 3,200 Ms. GW has a four-year-old son, who attends a day care center while she is at...
-
Answer the following question: The SWOT analysis compares internal strengths and weaknesses with external opportunities and threats so that management identifies areas of concern and opportunity both...
-
Assume Phipps Chips, a fictious snack company, makes artisan potato chips in a variety of flavors. The company sells 12-ounce bags of a variety of flavors in grocery store chains such as Whole Foods...
-
With the growing complexity of managing worldwide supply chains, coupled with significant pressures to reduce cost at the firm level, supply managers must learn to manage the inherent risks of global...
-
CVS Case Discussion In 2002, CVS assembled a team of operations executives and managers for its Pharmacy Service Initiative (PSI). Although CVS was one of America's largest retail drugstores,...
-
Imagine you are attending a conference on psychotherapy. A therapist presents findings about a new form of therapy that has been developed and tested in a small sample of people. Patients' scores on...
-
What If the Facts Were Different? Suppose that Morens predominant motive in bringing her son to the restaurant had been to benefit herself by feeding him free pizza. Would the result have been...
-
What tools are available to help shoppers compare prices, features, and values and check other shoppers opinions?
-
The uncertainty principle has negligible consequences for macroscopic objects. However, the properties of nanoparticles, which have dimensions ranging from a few to several hundred nanometers, may be...
-
The energy levels of a particle of mass m in a two-dimensional square box of side L are given by (n 1 2 + n 2 2 ) h 2 /8 mL 2 . Do any of these levels have the same energy? If so, find the values of...
-
Which of the following happens when the frequency of electromagnetic radiation decreases? Explain your reasoning. (a) The speed of the radiation decreases. (b) The wavelength of the radiation...
-
List of Accounts Sales Revenue 6 , 3 0 0 , 0 0 0 Income Tax Expense 2 0 % Interest expense 2 1 , 0 0 0 Cost of Goods Sold 4 , 1 0 0 , 0 0 0 Marketing, administrative, and other expenses 1 9 3 , 0 0 0...
-
On December 31, Hawkin's records show the following accounts. Cash Accounts Receivable $ 6,100 1,100 Supplies 4,000 Equipment 15,000 Accounts Payable 6,500 Common stock 11,900 Retained earnings,...
-
x and... ad Current Attempt in Progress This information relates to Sandhill Co. for the year 2022. Retained earnings, January 1, 2022 $95,100 Advertising expense 2,560 Dividends 8.520 Rent expense...

Study smarter with the SolutionInn App


