Which of the following isomers of diaminobenzene can form chelating complexes? Explain your reasoning. (a) NH NH,
Question:
Which of the following isomers of diaminobenzene can form chelating complexes? Explain your reasoning.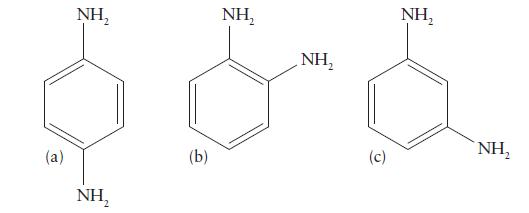
Transcribed Image Text:
(a) NH₂ NH, (b) NH₂ NH₂ (c) NH₂ NH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Only the molecule b can function as ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
In which of the following complexes are geometric isomers possible? If isomers are possible, draw their structures and label them as cis or trans, or as fac or mer. (a) [Co(H 2 O) 4 Cl 2 ] + (b)...
-
1. Hannah is applying for a life policy on her girlfriend Sarahs life. The policy is $500,000 and carries a large premium. Hannah is the main earner, so she is concerned about not being able to pay...
-
The multiple choice questions presented in Question 2 above are low context. You should be able to answer them simply by a careful reading of the chapter. These questions, on the other hand, are high...
-
Find the struggles faced by business (printed t-shirts) with suppliers? Explain what would be the communication plan for each stakeholder and how you'll implement the decision you have taken?
-
Assume you have the following jobs to execute with one processor: Suppose a system uses RR scheduling with a quantum of 15. a. Create a Gantt chart illustrating the execution of these processes. b....
-
Interstate Delivery Service is owned and operated by Katie Wyer. The following selected transactions were completed by Interstate Delivery Service during May: 1. Received cash in exchange for capital...
-
Students applying to graduate schools in many disciplines are required to take the Graduate Record Examination (GRE); essentially, it is the graduate school equivalent of the SAT. Let's say an...
-
Comprehensive Income Armstrong Corporation reported the following for 2010: net sales $1,200,000 cost of goods sold $720,000: selling and administrative expenses $320,000: and an unrealized holding...
-
The following data were gathered to use in reconciling the bank account of Bradford Company: Balance per bank $ 18,050 Balance per company records 10,040 Bank service charges 50 Deposit in transit...
-
Solutions of the [V(OH 2 ) 6 ] 2+ ion are lilac in color and absorb light of wavelength 806 nm. What is the ligand field splitting in the complex in kilojoules per mole?
-
What is the oxidation number of (a) V in VO 2+ ; (b) Zn in [Zn(OH) 4 ] 2 ?
-
a. Contrast inverse square law attenuation and absorptive attenuation. 8a.) What propagation problem is especially bad in moist air? a. Absorptive attenuation b. Electromagnetic interference c. Multi...
-
Find the moment at point A that keeps the given structure below in equilibrium in its given configuration using Virtual Work principle. 10kN B 2m 2m 2m 30 kN M 2m 2m 2m 30 A
-
The standard form of second order linear nonhomogeneous differential equation with constant coeffi- cients is given by (A) y"(x)+ Py'(x) + Qy(x) = R, where P,Q and R are functions of y or constants...
-
figure. The. Two converging water jets merge to create a unified, as illustrated in the While each individual stream undergoes a change in momentum upon collision, the total momentum of the entire...
-
search Question 12 of 15 -/1 3 Dorothy Taylor, a junior accountant for Sandhill Company, has been trying to determine the correct balance sheet for the company but can't seem to get it right. Her...
-
With the aid of diagrams, explain how an expansionary monetary policy is transmitted through an economy.
-
Kitchens are generally becoming smaller in relation to dining areas. Why?
-
Refer to the data for problem 13-36 regarding Long Beach Pharmaceutical Company. Required: Compute each division's residual income for the year under each of the following assumptions about the...
-
Although the vibrational degrees of freedom are generally not in the high-T limit, is the vibrational partition function evaluated by discrete summation?
-
What is the form of the total vibrational partition function for a polyatomic molecule?
-
How does the presence of degeneracy affect the form of the total vibrational partition function?
-
Provide a detailed marketing plan for International Delight coffee creamers.
-
CASE STUDY #1) Gaming market in LatAm (10/20 of the final grade) From: https://blog.boacompra.com/online-gaming-in-latam-market-stats/ There is no debate about the gaming industry's tremendous growth...
-
What role do emerging technologies, such as artificial intelligence, machine learning, blockchain, and augmented reality, play in shaping the future of organizational development, and how can...

Study smarter with the SolutionInn App


