Write the chemical formula for the conjugate base of formic acid, HCOOH and calculate its pK b
Question:
Write the chemical formula for the conjugate base of formic acid, HCOOH and calculate its pKb from the pKa of formic acid (see Table 6C.1).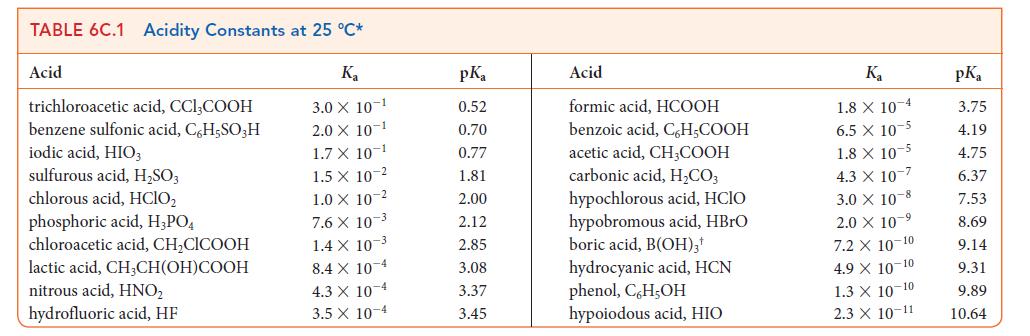
Transcribed Image Text:
TABLE 6C.1 Acidity Constants at 25 °C* K₂ 3.0 X 10-1 2.0 X 10-1 1.7 X 10-1 1.5 X 1.0 X 10-2 Acid trichloroacetic acid, CCI,COOH benzene sulfonic acid, C6H5SO3H iodic acid, HIO3 sulfurous acid, H₂SO3 chlorous acid, HClO₂ phosphoric acid, H3PO4 chloroacetic acid, CH₂CICOOH lactic acid, CH3CH(OH)COOH nitrous acid, HNO₂ hydrofluoric acid, HF 10 2 7.6 X 10-3 1.4 X 10-3 8.4 X 10 4 4.3 X 10-4 3.5 x 10-4 pKa 0.52 0.70 0.77 1.81 2.00 2.12 2.85 3.08 3.37 3.45 Acid formic acid, HCOOH benzoic acid, C,H₂COOH acetic acid, CH,COOH carbonic acid, H₂CO3 hypochlorous acid, HCIO hypobromous acid, HBrO boric acid, B(OH)3¹ hydrocyanic acid, HCN phenol, C,H,OH hypoiodous acid, HIO K₂ 1.8 X 10 4 6.5 x 10-5 1.8 X 10-5 4.3 X 107 3.0 X 108 2.0 × 10-⁹ 7.2 X 10-10 4.9 X 10-10 1.3 X 10-10 2.3 X 10-11 pKa 3.75 4.19 4.75 6.37 7.53 8.69 9.14 9.31 9.89 10.64
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
HCO pK...View the full answer

Answered By

Muhammad Haroon
More than 3 years experience in teaching undergraduate and graduate level courses which includes Object Oriented Programming, Data Structures, Algorithms, Database Systems, Theory of Automata, Theory of Computation, Database Administration, Web Technologies etc.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
1. Does squinting help you see things more clearly? Explain. 2. Why, when there are very low-light conditions, are you unable to see color? 3. Stand in a well-lit room facing a mirror. Close your...
-
Find α and β that solve the following equation: 1 2 2 1
-
In order to have $391,185 in 26 years, how much needs to be deposited each month into a bank account whose annual rate is 1.8% with monthly compounding?
-
Standard-costing method, assigning costs. Refer to the information in Exercise 17-24. Suppose Bio Dec determines standard costs of $6.60 per equivalent unit for direct materials and $10.40 per...
-
The following payoff table provides profits based on various possible decision alternatives and various levels of demand at Amber Gardners software firm: The probability of low demand is 0.4, whereas...
-
What does a class diagram portray?
-
A bank issues a $100,000 fixed-rate 30-year mortgage with a nominal annual rate of 4.5%. If the required rate drops to 4.0% immediately after the mortgage is issued, what is the impact on the value...
-
Following are the questions of case study: Question:1 What is the evaluation you need to make OR decision you need to make OR problem you need to diagnose? Answer should be clear (easy to read - 1...
-
Identify (a) The Brnsted acid and base in the following reaction, and (b) The conjugate base and acid formed: HNO3(aq) + HPO (aq) NO3 (aq) + HPO4 (aq)
-
Calcium acetate, Ca(CH 3 CO 2 ) 2 (aq), is used to treat patients with a kidney disease that results in high levels of phosphate ions in the blood. The calcium binds to the phosphates so that they...
-
From this equation you know that an increase of 1 unit in FBS is associated with what change in HbA1c? Khan, Sobki, and Alhomida (2015) examined 75 patients to assess the association between fasting...
-
Calculate Accrued Interests for Each Bond by the Date of 01/01/2023 Calculate the price for the Bonds for the First day of every year till Maturity Year Calculate Macaulay's Duration and Modified...
-
First identify and label any variable(s) in this problem. (Diagram not drawn to scale.) Then write an equation(s) and show how to solve it algebraically. Be prepared to show your work. Cheryl fenced...
-
Simplify it (6-8 - + +8) sJ+ "
-
On march 15, max invested cash in an amount equal to the current market value of Weber's partnership captit. The partners decided that max would earn 70 percent of the partnership profits because...
-
A temperature field T(x,t) in a one-dimensional structure (x = [0, L]) is determined by 1 ar 8T the following governing equation Based on previous experience, one a t proposes that the temperature...
-
How does the problem of managing companies resemble the problem a pilot faces when flying a jetliner?
-
Write an SQL statement to display all data on products having a QuantityOnHand greater than 0.
-
Show that the three wave functions in Equation (14.11) are normalized.
-
Discuss why a quantum mechanical particle in a box has zero point energy in terms of its wavelength.
-
How does an expectation value for an observable differ from an average of all possible eigenvalues?
-
Draw your solution to part a on a separate piece of paper (to be submitted on eLearning along with all other handwritten work). Submit your answer to part b here. a. Draw the circuit represented by...
-
The purpose of this exercise is to provide the experience of creating interactive software by using Python. The assessment contributes to all learning outcomes as indicated and will comprise of the...
-
5) You have made one straight-through patch cable ~ 3ft as below (T568A Color). Briefly explain the steps involved in terminating both ends. (10 points) Connector Head Bottom Side Up #T568A COLOR 1...

Study smarter with the SolutionInn App


