You may need Table 7.2 to answer the following questions. a. Which is the stronger base, Cl
Question:
You may need Table 7.2 to answer the following questions.
a. Which is the stronger base, Cl2 or H2O?
b. Which is the stronger base, H2O or NO22?
c. Which is the stronger base, CN2 or OC6H5?
Table 7.2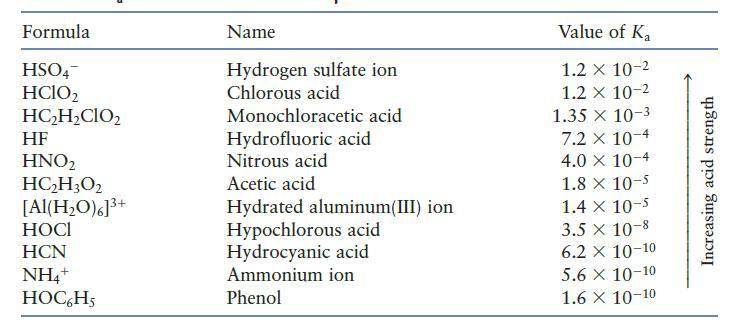
Transcribed Image Text:
Formula HSO4 HClO2 HC₂H₂CIO₂ HF HNO₂ HC₂H3O2 [Al(H₂O)6]³+ HOCI HCN NH4+ HOC6H5 Name Hydrogen sulfate ion Chlorous acid Monochloracetic acid Hydrofluoric acid Nitrous acid Acetic acid Hydrated aluminum(III) ion Hypochlorous acid Hydrocyanic acid Ammonium ion Phenol Value of Ka 1.2 x 10-2 1.2 x 10-2 1.35 x 10-3 7.2 x 10-4 4.0 X 10-4 1.8 x 10-5 1.4 x 10-5 3.5 x 10-8 6.2 X 10-10 5.6 X 10-10 1.6 X 10-10 Increasing acid strength
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 27% (11 reviews)
a The pKa of Cl2 is 36 while the pKa of H2O is 1574 Since Cl2 has ...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Answer the following questions about internal control over cash payments: 1. Payment by check carries three controls over cash. What are they? 2. Suppose a purchasing agent receives the goods that he...
-
Answer the following questions based on Tables 5P-1 and 5P-2. a. What is the quantity demanded at $10? What is the quantity supplied at $10? b. What is the quantity demanded at $25? What is the...
-
Answer the following questions based on two assumptions: (1) Inflation increases the prices of all goods by 20%. (2) Ina's income increases from $50,000 to $55,000. a. Has Ina's budget line become...
-
How can staff review the effectiveness of their work, the services they provide and the social and cultural factors impacting on clients, groups or communities?
-
Suppose that X and Y are independent random variables. Suppose that X has a discrete distribution concentrated on finitely many distinct values with p.f. f1. Suppose that Y has a continuous...
-
Evan Company operates three segments. Income statements for the segments imply that Evan could improve profitability if Segment X is eliminated. Required a. Explain the effect on Evan's profitability...
-
Given two rectangular ducts with equal cross-sectional area, but different aspect ratios (width/height) of 2 and 4 , which will have the greater frictional losses? Explain your answer.
-
Harrison Company makes two products and uses a traditional costing system in which a single plantwide predetermined overhead rate is computed based on direct labor-hours. Data for the two products...
-
Discuss the cultural differences between China and the United States. What would be the biggest adjustments I might need to make if working there as an expatriate? What are the most...
-
Use Venn diagrams to evaluate the immediate inferences in Part II of this exercise. Identify any that commit the existential fallacy. In part 1. No sculptures by Rodin are boring creations....
-
Write balanced equations that describe the following reactions. a. The dissociation of perchloric acid in water. b. The dissociation of propanoic acid (CH 3 CH 2 CO 2 H) in water. c. The dissociation...
-
Consider the following statements. Write out an example reaction and K expression that are associated with each statement. a. The autoionization of water. b. An acid reacts with water to produce the...
-
The auditor of a company is required to form and express an opinion on the truth and fairness of the companys financial statements. Explain briefly: (i) The meaning frequently given to the phrase...
-
Griffin buys and sells ladies handbags. The average selling price is $ 4 1 0 per bag. Average variable costs ( purchase price of bags plus freight costs and selling expenses ) amounts to $ 1 9 0 per...
-
Saleena was hired and agreed to a $48.50 per hour compensation, for a regular 44 hour work week. Calculate her net earnings for the year. (10 marks) Federal Tax Brackets In Canada 2022 Federal Tax...
-
What quality assurance protocols do you employ to test and validate data integrity in software applications and databases ?
-
A company pays $ 5 , 0 0 0 for equipment. Annual depreciation on the equipment is $ 5 0 0 . What is the book value of the equipment at the end of Year 2 ?
-
a. Equipment depreciation was $1,500. b. $700 of advertising expense was incurred but not paid. (Use Advertising Payable.) c. Office Supplies on hand at the end of the year totaled $250. The...
-
a) Derive planar density expressions for BCC (100) and (110) planes in terms of the atomic radius R. (b) Compute and compare planar density values for these same two planes for vanadium.
-
What are the three kinds of research types? Explain each type.
-
What volume of 0.100 M Na 3 PO 4 is required to precipitate all of the lead(II) ions from 150.0 mL of 0.250 M Pb(NO 3 ) 2 ?
-
A 1.00-g sample of an alkaline earth metal chloride is treated with excess silver nitrate. All of the chloride is recovered as 1.38 g of silver chloride. Identify the metal.
-
A mixture contains only NaCl and Al 2 (SO 4 ) 3 . A 1.45- g sample of the mixture is dissolved in water, and an excess of NaOH is added, producing a precipitate of Al(OH) 3 . The precipitate is...
-
What is the discounted rate of Stellantis NV ? Please calculate the discounted cash flow od Stellantis using the income approach.
-
7- Suppose that you are editing a file that contains the following paragraph and the cursor is on the second tilde (~): The vim editor has a command, tilde (~), that changes lowercase letters to...
-
A $1,000 par value bond has a 12% coupon rate (paid annually). It has 10 years remaining to maturity. If bonds of similar risk and maturity currently yield 8%, what should this bond's price be?

Study smarter with the SolutionInn App


