Fill in the blanks in the table. Relief configuration a. Single relief, nonfire b. Single relief, nonfire
Question:
Fill in the blanks in the table.
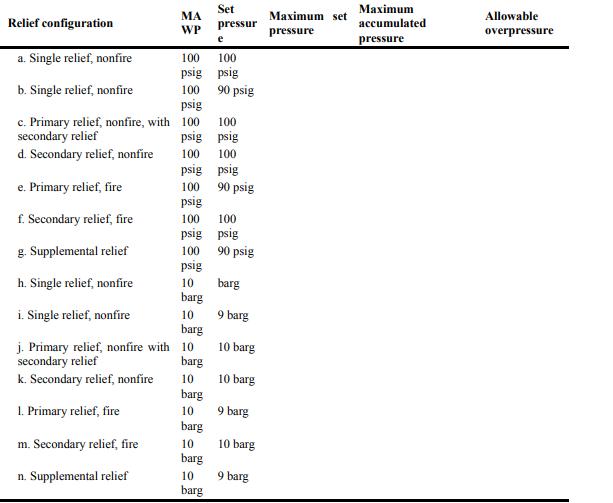
Transcribed Image Text:
Relief configuration a. Single relief, nonfire b. Single relief, nonfire c. Primary relief, nonfire, with secondary relief d. Secondary relief, nonfire e. Primary relief, fire f. Secondary relief, fire g. Supplemental relief h. Single relief, nonfire i. Single relief, nonfire j. Primary relief, nonfire with secondary relief k. Secondary relief, nonfire 1. Primary relief, fire m. Secondary relief, fire n. Supplemental relief MA WP 100 100 psig psig 100 90 psig psig 100 100 psig psig 100 psig 90 psig 100 psig 100 psig Set pressur 100 100 psig psig 100 90 psig psig 10 barg 10 barg 10 barg 10 barg 10 barg 10 barg 10 barg barg 9 barg 10 barg 10 barg 9 barg 10 barg 9 barg Maximum set pressure Maximum accumulated pressure Allowable overpressure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)

Answered By

Rajat Gupta
used to take tution classes from my school time.
Conducted special topic claases during my graduation to help the students pass their exams.
Currently, teaching and conducting online claases during my post- graduation too.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemical Process Safety Fundamentals With Applications
ISBN: 9780134857770
4th Edition
Authors: Daniel A. Crowl, Joseph F. Louvar
Question Posted:
Students also viewed these Engineering questions
-
Fill in the blanks in Table 5-12. FUNCTIONAL FORMS OF REGRESSION MODELS Model When appropriate Yi = B1 + B2 (i)
-
Fill in the blanks in the following table of GDPstatistics: 2005 2006 2007 13,843 Nominal GDP 12,434 Real GDP 11,003 11,319 GDP deflator 116.6 119.7
-
Fill in the blanks in the following table with the major product(s) of the reaction of each haloalkane with the reagents shown. Reagent KOC(CH,) Haloalkane CH C CH,CH,CH2CI (CH3)2CHCI CH2CCI H20 H,O...
-
Draw a UML Sequence Diagram for the process involved in paying a vendor for an item in cash. The process to follow is below. [15 points] a) Go to the bank b) Request your account balance c) The bank...
-
Consider the composite body shown. Determine (a) The value of x when h = L/2, (b) The ratio h/L for which x = L.
-
What are some of the advantages in using a predetermined time system as opposed to stopwatch time study?
-
Is there a relationship between the number of accounting standards issued and demand for accounting and auditing services? Explain this relationship. Given this relationship, should environmental...
-
For what purposes are departmental reports useful to management?
-
Your tasks are: 1A) Generate active portfolio A (report weights wA1, wA2, wA3, and wA4) 2 1B) Generate key parameters of portfolio A (report A, BA, oA; RA, oA, Cov(RA, RM)) 1C) Use the information...
-
Calculate the diameter of a certified capacity spring-type liquid relief for the following conditions: Pump capacity at Ap Set pressure a. 100 gpm 50 psig 100 psig b. 200 gpm 5 m/s 5 barg C. d. 7 m/s...
-
Decide which types of relief devices and configuration should be used for the following situations. Explain. a. The process fluid is highly toxic. b. The process operates close to the MAWP of the...
-
A $15,000 bond with a contract interest rate of 6% was issued on March 1, 2017. Calculate the cash paid on the first interest payment date if interest is paid: a. Annually b. Semiannually c....
-
Suppose joel decides to go fishing at Lake Veronica on his day off from work, but he faces a choice: he can fish the north, south, east, or west side of the lake. His expected catch for the day is as...
-
If the Sun replaced by the star Rigil Kentaurus, how many times greater is the power received from Rigil Kentaurus compare to the power received from the Sun?
-
How do emerging technologies such as machine learning and artificial intelligence facilitate predictive modeling and early detection of potential process safety incidents? Explain
-
Simplify: 523.515 Note: Do not evaluate. Provide your answer below:
-
How do inherent safety principles influence the design and operation of chemical processes to mitigate potential hazards ?
-
On February 1, 2013, Garzon purchased 6% bonds issued by PBS Utilities at a cost of $ 40,000, which is their par value. The bonds pay interest semiannually on July 31 and January 31. For 2013,...
-
Hardin Services Co. experienced the following events in 2016: 1. Provided services on account. 2. Collected cash for accounts receivable. 3. Attempted to collect an account and, when unsuccessful,...
-
(a) What value of absorbance corresponds to 45.0% T? (b) If a 0.010 0 M solution exhibits 45.0% T at some wavelength, what will be the percent transmittance for a 0.020 0 M solution of the same...
-
(a) A 3.96 10 4 M solution of compound A exhibited an absorbance of 0.624 at 238 nm in a 1.000-cm cuvet; a blank solution containing only solvent had an absorbance of 0.029 at the same wavelength....
-
Ammonia can be determined spectrophotometrically by reaction with phenol in the presence of hypochlorite (OCl - ): 4.37-mg sample of protein was chemically digested to convert its nitrogen into...
-
Carla Vista's Custom Construction Company is considering three new projects, each requiring an equipment investment of $26.400. Each project will last for 3 years and produce the following net annual...
-
A single mass m 1 = 3.8 kg hangs from a spring in a motionless elevator. The spring is extended x = 10 cm from its unstretched length. a. What is the spring constant of the spring? Now, three masses...
-
What is a share class? What is a prospectus? What is a stock index? What is the Dow Jones Industrial Average? What is the S&P 500? What is the NASDAQ? What is a stock split? What is a reverse stock...

Study smarter with the SolutionInn App


